How FTIR works II
The Editor
Fabrice Birembaut*
*University of Southampton
Highfield, Southampton
SO17 1BJ
UK
In this piece we will attempt to explain the enormous range of software options you have available on even the simplest FTIR. If you go to your instrument and open up the settings available, you will invariably find “resolution”, “number of scans” and “background parameters”, all of which were discussed in the first article [reference] and then a whole mass of confusing options such as “Apodization functions”. In addition you are sometimes offered the facilities to include “Zero filling” and its hard work to find out what they all do. Just to confuse you further, you are offered a set of data ‘improvement’ procedures including “averaging” and “KK processing” and often lot’s more. In our explanation of some of these below, we assume you will read the article at your FTIR and play games on your own instrument to demonstrate to yourself what each function does. In all cases, we are going to use the standard polystyrene test spectrum so we assume you have a thin film of polystyrene available. [Most instrument manufacturers provide these when they deliver the instrument]. If you don’t have a film – despair not. Stand a transparent polystyrene ball pen in a small test tube of chloroform until it goes gooey (about 10 minutes). Stir it around and then pipette a generous coating of the chloroform solution onto a horizontal polished KBr flat. Allow it to dry in a fume hood (keeping the flat just a little warm helps). Put the coated flat in your FTIR and run a spectrum and you should see something like this –
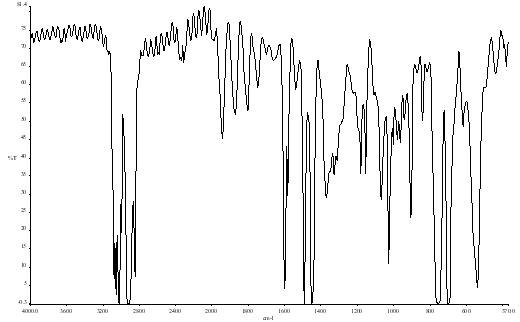
Figure 1. Polystyrene Film Spectrum
Background Resolution: 4 cm-1 # of scans. 4
Spectrum Resolution: 4 cm-1 # of scans. 4
Apodization Functions
When your FTIR scans, it actually records an interferogram, which is subsequently digitised, and then Fourier Transformed into a spectrum (See Article 1 for details).
The form of the interferogram is
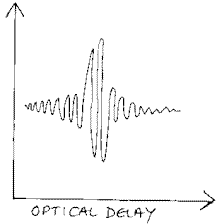
The vertical axis is never labelled i.e. it is in arbitrary units BUT in reality it is the output voltage or current of the detector. Unlike a spectrum, which shows peaks against a background, the interferogram shows swings of output about a non-zero mean. At the end of each scan, the machine reverses its direction. Inevitably the reversal means that the scan must be slowed down, stopped, reversed and then sped up again. As the optical delay is slowed, stopped and then accelerated, the output data is corrupted, so the instrument is told to switch off the detector until the reversal is complete and the movement has become nice and smooth. So to put it diagrammatically we have –
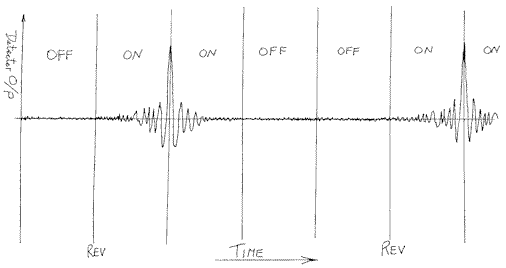
Figure 2.
And when you display the interferogram you only ‘see’ the good bit, the span between the two switching intervals.
Fine – but there is a problem. If you switch off a signal, it will naturally fall rapidly to zero i.e. there will be a ‘step’ in the output value of the detector circuit. Unfortunately, the Fourier Transform processor will process this step and will make spectral nonsense out of it. The trick is to minimise this nuisance by lowering the output along a chosen curve – an “apodization function”. Each function has a different shape eg.,

Figure 3.
To demonstrate the effect of these, we recorded spectra of polystyrene under various conditions and compared them. All spectra were recorded with a TGS detector at 4cm-1 using 4 co-added scans. The backgrounds and the spectra were each recorded under identical conditions.
| Compare | Spectrum 1 | Strong (Beer Norton) Apodization | See Figure 1 |
| Spectrum 2 | Weak apodization | See Figure 4a and 4b | |
| Spectrum 3 | Boxcar – no apodization | See Figure 5a and 5b |
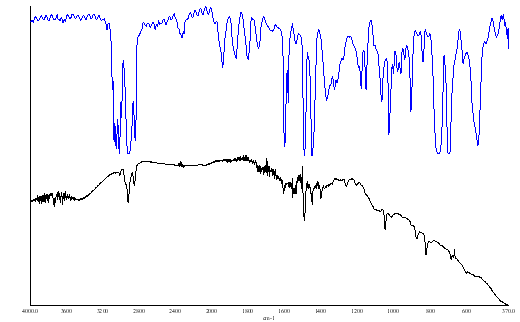
Figure 4. (a) background (b) PS film both with a weak apodization
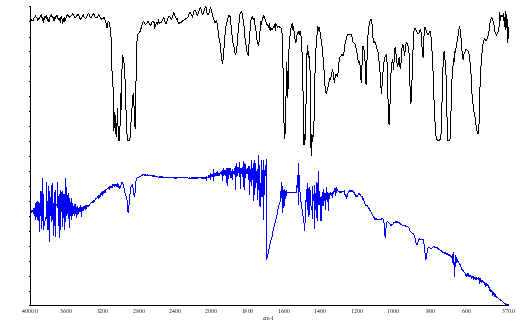
Figure 5. (a) background (b) PS film both with a boxcar apodization
The spectra of polystyrene showed little or no effect as one changes the apodization however, where bands are sharp one can see effects. Look at the backgrounds and you will see that reducing or switching off the apodization can cause the sharp vibration rotation bands of water vapour to alter in apparent position and shape. So – if you are going to record spectra of gases especially at higher resolutions or of crystalline solids with very sharp bands you must be careful about apodization. If too low the bands will appear to be better resolved but ‘ringing’ can occur.
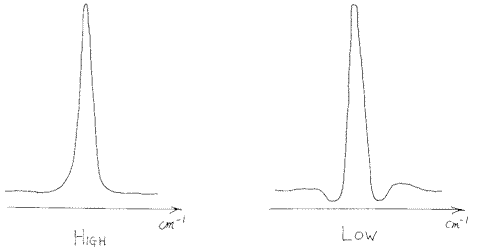
Figure 6.
In most analytical spectroscopy there is little problem. Since the bands are broad, the effect of using high apodization is not disastrous and it is safer to use it rather than not.
Smoothing
Older spectrometers offered simple smoothing procedures eg., the instrument took 3, 5 or even more spectral data points, averaged them and plotted out the average. It then moved by one cm-1 increment (usually ½ resolution value) and repeated. These days more sophisticated functions are used but the effect is similar – you reduce the noise by averaging but unfortunately you lose resolution.
Let’s try an experiment. Take a piece of Al foil (kitchen quality is just fine) and punch a hole in it 1 to 1.5mm in diameter with a needle of sharp pencil. Place this over your polystyrene film and put the sandwich in the spectrometer after you have run a background. Make sure the hole is roughly in the middle of the IR beam. Run a spectrum. You should see something like this –
Figure 7.
That is the whole spectrum is squashed up – very little light passes the hole in the foil. Now expand the spectrum to fill the screen – Figure 8.
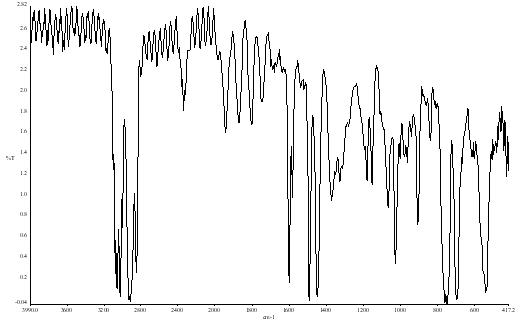
Figure 8.
The spectrum is fine but the result is noisy. This problem can arise where for some reason the energy is very restricted (examples include the use of some spectral reflection techniques, spectra on samples held in some high or low temperature cells or in IR microscopy). The correct way to deal with it is to co-add many scans [Remember, the S:N ratio – improves as v# of scans]. Either this may not be possible or just take too long, or the experiment is finished and you want to clean up the results you have and not do the job again. The application of smoothing is worth considering.
In Figure 9 we show the effect of smoothing the data in Figure 8.
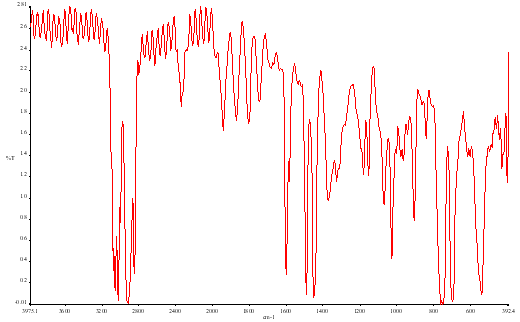
Figure 9.
There is a loss of resolution but the noise is indeed reduced. In this case the loss of resolution is acceptable but in some other cases it may not be.
Absorbance vs Transmittance
In our experience most analytical users of infrared record spectra in transmission (0-100% transmission) and never use absorbance. The theory behind this is –
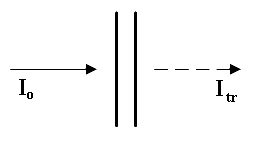 Transmittance = Itr x 100%
Transmittance = Itr x 100%
Io
Absorbance = –Loge Itr
Io
or if you prefer = – ln Itr
Io
The negative sign means that the peaks in Absorbance are the negative ones in transmission.
In Figure 10 we show part of the spectrum of polystyrene presented in transmission (bands downwards) and absorbance (bands upwards).
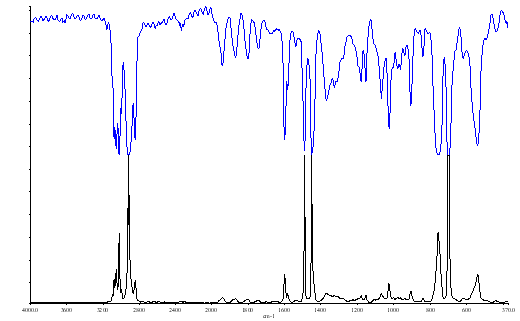
Figure 10.
Several points are clear –
- Absorbance spectra look much better than transmittance. The bands appear to be sharper and clearer.
- The width at half height W½ is narrow in transmission.
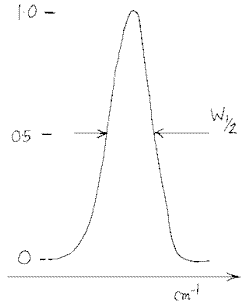
The band at 1451 cm-1 in polystyrene has W½ of ~18cm-1 in transmittance and ~2½cm-1 in absorbance. If you check the vibration rotation gas phase spectrum of water vapour in the background you will find less of an effect eg., the band at 1050 cm-1 narrowed from W½ = 6.5 to about 5cm-1 .
So the question must arise – why not use absorbance more often as a method of presentation.
You will note that in Figure 10b, the absorbance scale goes from 0 to 7 i.e. 100 to about 0.1%.
This article is getting a bit long so we will prepare How FTIR Works III for you and include differentiation and other parameters
REF: P.J. Hendra & F. Birembaut, Int. J. Vib. Spect., [www.irdg.org/ijvs] 6, 2, 3 (2002)