Polarised light in the mid infrared
The Editor
Relatively few users of infrared have considered the use of polarised light. In a sense, this is a little odd because in some areas of optical science, polarisers are considered to be essential equipment. Thus, optical microscopy is almost always associated with the use of polarised light either in transmission or reflection. Petrographic microscopy on mineral sections, fracture surfaces in engineering – all use polarised light to improve the resolution or to enhance the analysis. Polarisers in infrared analysis are also really valuable but rarely used – Why?
Background principles
If a sample is oriented, specific vibrations have a directional component. Let us consider nylon as an example. An oriented film of nylon will have its molecular axes along the stretch (deformation) direction. The orientation will not be perfect but it certainly will be preferred.
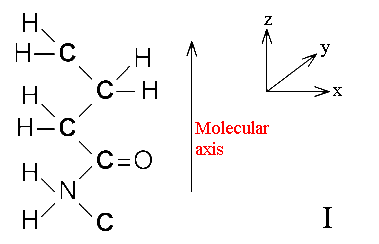
Quite obviously those vibrations of this molecule that have vectors predominantly along the chain will interact best with resonant infrared radiation polarised vertically. On the other hand, the C=O stretching mode has a vector perpendicular to this direction.
In a polymer there is almost always only one preferred direction – the stretch direction i.e. the chain axis. In the diagram above, the z axis lies along the chain but x + y have no significance.
There are exceptions – many examples of polyethylene terepthalate (PET) are 2 way oriented. In this case stretching occurs in two orthogonal directions and hence as in a crystal, directions x, y + z each have significance. Coke bottles are just such a case. A small ‘pre-form’ with a neck of the finished size and shape is gripped at the neck, the body of the pre-form is heated and then blown with hot air into a mould of size identical to the finished product. The PET sheet forming the walls deforms in 2 directions and hence the benzene rings lie roughly parallel to the wall surfaces.
There are many other examples of oriented materials from minerals to fibres, films, some coatings, liquid crystals etc, etc. Orientation can even be present and be an unwanted nuisance. In all these cases infrared can be pressed into service.
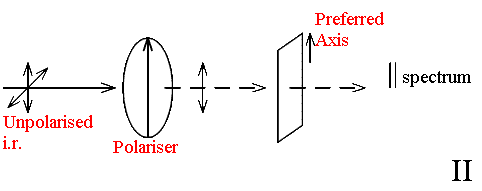
The principle is to introduce a polariser into the optical system and study the spectrum with the sample oriented in different ways with respect to the polariser.
In the case of a film or fibre two measurements are obvious.
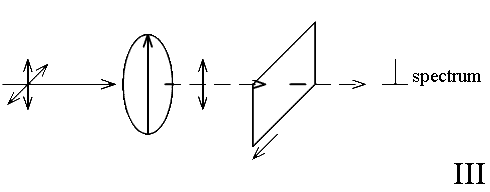
If 3 way orientation is to be monitored a third measurement can be made.
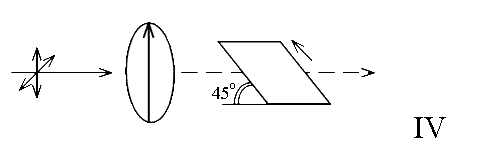
i.e. the film is inclined with respect to the beam at 45º.
In II the spectrum will be predominantly that of modes with vectors parallel to the molecular axis whilst the – spectrum will emphasise those vibrating across the chain.
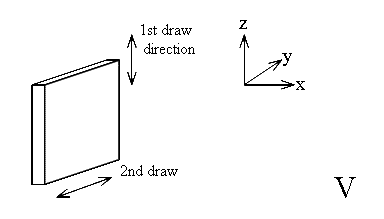
In the 3 way oriented case we will set x as the out-of-plane axis. If the sample is PET the out of plane CH deformations of the aromatic rings will lie along the axis.
The third experiment shown in diagram IV attempts to exploit this. The method goes back to Harry Willis’s pioneering work on the vibrational spectra of polymers at ICI in Welwyn Garden City, UK. The amount of interaction between the vector and the polarisation direction varies as CosΘ
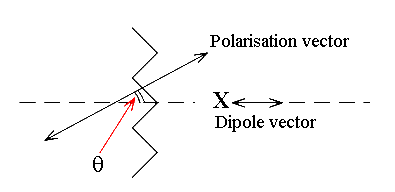
If the angle of inclination is known in experiment IV and it is, the component in the spectrum due to the contribution of modes to dipole vectors along axis x can be unscrambled from those vibrating along y and z.
Experimental
Perhaps there are two reasons why people so rarely use polarised light –
- the polarisers themselves and
- nervousness that reproducible meaningful results can be generated.
Over the years, a wide range of polarisers has been used. Early ones were based on stacks of sheets of optically transmission material set at Brewter’s angle to the beam. Silver chloride was popular when I was a student. These polarisers were very clumsy and were replaced by “wire grid” devices many years ago. These consist of a piece of optical window (usually ZnSe these days) vacuum coated with a gold grid consisting of fine gold strips separated by gaps narrower than the radiation to be passed through the device. These polarisers are highly efficient but both expensive (~$1500E) and extremely fragile. Touching the gold wire grid with anything will wipe off the gold and ruin the device.
Some manufacturers arrange a polariser stand or filter wheel inside the sealed part of the system enabling the user to insert and remove the polariser under software control. This is ideal – no-one other than the service engineer will touch the polariser surface and it stays clean, dry and free from dust.
Where such a facility is not available, the user must position the polariser in the sample area. Suppliers of polarisers usually mount them in a ring (often 25mm in diameter and ~5mm thick). They invariably provide a mark or dot to indicate the electric vector direction. To use the polariser in the sample area it is not adequate to tape it into position or prop the thing on a lump of blue tack or play dough. No-you need a mount, I draw below one we had at Southampton – very simple, completely effective and it held the precious polariser firmly and safely.
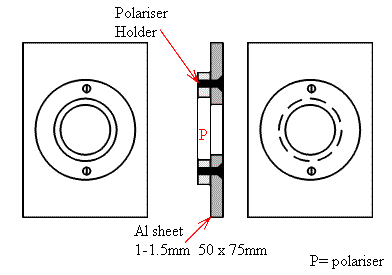
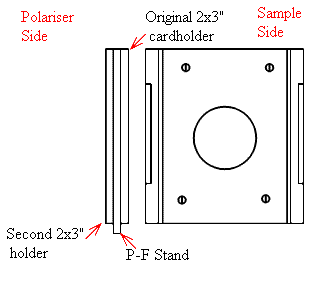
Second Holder retained using 2×3″ strips of double-sided sticky tape.
Hole cleaned out with a sharp knife.
As I have shown in diagrams II and III we need two spectra in orthogonal directions. It would appear to be simplest to rotate the polariser and leave the sample fixed especially if experiment V is to be included but no – the rule is, leave the polariser fixed and rotate the specimen. The reason is that diffraction instruments and interferometers are themselves polarising filters # i.e. they have a preferred orientation and the preference changes with wave length (cm-1). In an interferometer, the trick is to set the polariser usually with the vector vertical or horizontal, run the background through the polariser and use this for all subsequent experiments. Some experts prefer the set the polariser tilted at 45º to the vertical, but this can cause confusion and mistakes in use.
# Interferometers are usually for less polarising than diffraction instruments. In some instruments the transmission is almost identical in horizontal and vertical directions but it is unsafe to rely on this.
ATR
Years ago, when Harry Willis was a regular visitor to my group at Southampton, he got me to set up an ATR system based on a truncated square pyramid crystal. The idea was to clamp the sample on one of the flat surfaces, then run the polarised spectrum and rotate the sample and crystal sandwich by 90º before repeating.
I wondered more recently if diamond ATR would work with a polariser. The answer somewhat to my surprise is ‘yes’!
Again, the polariser is inserted in the instrument and left fixed, the sample is placed in the diamond ATR either parallel or perpendicular to the beam direction. In the figure below a polypropylene filament was used. The two experiments were –
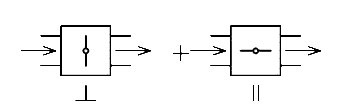
You can see the effect very well – some bands are stronger in the II spectrum, others in the ? spectrum. The perfection or degree of orientation is related to this dichroism as it is called

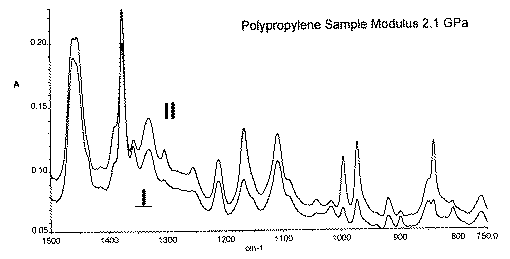 For some polymer applications it is essential to monitor the degree of orientation because it is a first class indicator of the mechanical performance of the material.
For some polymer applications it is essential to monitor the degree of orientation because it is a first class indicator of the mechanical performance of the material.
Fine I hear you say, but in diamond ATR the sample is crushed onto the horizontal surface of the diamond. Surely any measurement made on such a mangled surface is hardly typical of the real material. I disagree – it is well known that oriented materials are either fibrillar or behave as such. If you crush a fibrillar material e.g. timber, the material retains its orientations as it crushes and de-fibrillates.
Idea for a project
Many of the failures in deep vacuum formed products such as yoghurt pots arise at the changes of section. The bottoms may crack or the rim make break away. The problem arises from residual orientation – the sheet of polymer is particularly violently stretched at these points and if the polymer sheet is not adequately heated orientation can become ‘frozen in’ to the product. The simplest way of checking is to heat the product until it softens – the oriented molecules will then cause the product to shrink and/or distort. If a grid is printed onto the plastic before heating the contraction afterwards yields a measure of the orientation.
Infrared would be an ideal way to monitor the orientation in the suspect areas but until the development of polarised diamond ATR this was virtually impossible. The products are always far too thick to enable spectroscopists to use transmission unless the offending parts are microtained. Diamond ATR would seem to me to be ideal!
Has anyone tried?
Conclusion
Any method of increasing analytical data enhances the reliability or specificity of the analysis. As so many specimens have at least a small degree of orientation – a preferred direction, the use of polarised radiation will improve the analysis.
As I pointed out above, orientation even slight orientation can have serious consequences e.g. in service, long period distortion or enhanced risk of cracking or breakage in materials.
So – go and get a polariser and see what fun you can have. Only don’t touch the surface!
Editor
REF: P.J.Hendra, Int. J. Vib. Spect., [www.irdg.org/ijvs] 5, 1, 3 (2001)