Measurements of impurities in strongly absorbing powdery materials by DRIFTS
10. Measurements of impurities in strongly absorbing powdery materials by DRIFTS
Jimmy Bak
Optics and Fluid Dynamics Department,
Risø National Laboratory,
4000-Roskilde,
Denmark.
Abstract
Diffuse reflectance infrared Fourier transform spectrometry (DRIFTS) has been demonstrated to be capable to determining small concentrations down to the 100 ppm level (0.01 wt %), of strontianite (SrCO3) in powdery ceramic samples. Ceramic samples made of Bi-Sr-Cu-Ca-O (BiSCCO), which are used as superconducting precursors, absorbs strongly in the mid-infrared due to their semi-metallic properties. It is necessary, therefore, to dilute these powders down to a few wt % in order to obtain a linear relationship between absorbance and concentration values. A calibration approach based on the method of standard additions is employed in order to determine the SrCO3 concentrations in the contaminated BiSCCO powders.
Introduction
Diffuse Reflectance Infrared Fourier Transform Spectroscopy (DRIFTS) is a useful technique for measuring the amount of ionic compounds present in ceramic powders. The following advantages of using DRIFTS are:
- DRIFTS is a sufficiently sensitive technique in the Mid-IR spectral region, where ionic compounds such as: sulfates, nitrates, phosphates and carbonates absorbs strongly. Detection of ionic compounds in low concentrations is therefore possible.
- DRIFTS is a very fast and specific method for analyzing the ceramic powders for ionic compounds. Preparation of samples, recording of FTIR-spectra and data analysis can be carried out within an half an hour.
DRIFTS has been applied to the analysis of a variety of powdered materials, e.g.
- inorganics: coals1, cement2, kaolin clay3, silicon nitride4, calcium carbonate5 and superconductor precursor powders6 .
- organic materials: carbohydrates7 and caffeine1,8.
Quantifying the concentrations of the chemical compounds by infrared spectrometry in powders is, however, a much more difficult task than in gas and liquid analysis. It has been recognized, that parameters such as: particle size9, sample packing density8,10, optical properties of the powder11 all influence the measurements.
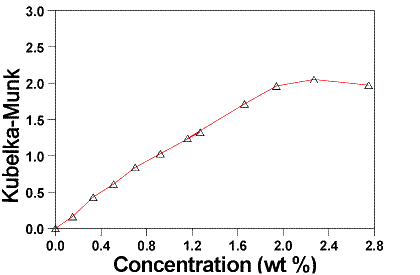
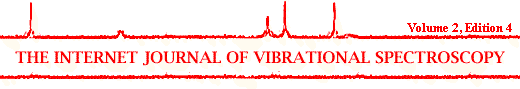
Figure 1.The measured spectra of the strongly absorbing BiSSCO powders expressed in KM values respond linearly at 1479.4cm-1 (carbonate peak) up to ~ 2 wt% only
In this work it is demonstrated that DRIFTS can be used to determine small amounts of strontianite (CaCO3) in ceramic powders which are strongly absorbing in the infrared spectral region. Materials which absorb strongly in major parts of the spectral region are troublesome when impurities are to be quantified. Looking at Figure 1 it is observed that the spectral response curve is only linear at low concentrations up to » 2.0 wt % of the absorbing material diluted in an infrared transparent matrix. The response curve is flat and constant from 2-100 wt %. It is necessary, therefore, to dilute the ceramic powders strongly before they are analyzed in order to be able to quantify the impurities. The experimental data is expressed in Kubelka-Munk (KM) units i.e.:
KM = f(R¥ ) = (1 – R¥ )2/2 R¥ = k/s
where R¥ = Rs/Rref is the ratio of the intensities of the IR radiation reflected from the surface of the ceramic powder diluted in KBr and that reflected from a non-absorbing KBr reference. f(R¥ ) is in many cases a linear function of concentration. A detailed discussion of the Kubelka-Munk theory is presented elsewhere.11,12
Experimental
The FTIR instrument used in all the experiments was a Bomem MB100. The spectral resolution of the interferometer was set at 4 cm-1 for quantitative analysis. A cosine apodization function was used for all the spectra. The interferometer was equipped with a standard DTGS detector. A Harrick “Praying Mantis” diffuse reflectance accessory with off-axis geometry was used in all the experiments to record the spectra of the powder materials.
Merck spectroscopy grade potassium bromide (KBr) was used both as a reference material for recording reference spectra and as a diluent. The ceramic powder samples of nominal composition Bi1.84 Pb0.34 Sr1.91 Ca2.03 Cu3.06 Ox containing small amounts of strontianite were produced in our laboratory. The SrCO3 (strontianite) used for the calibration work is a Merck product. A commercial Bi-Pb-Sr-Ca-Cu-O powder free of carbonates purchased at Merck was used to examine the sensitivity of the DRIFTS method.
A balance with an accuracy of 0.1 mg was used to weigh out all the samples diluted in dry KBr (0.7-1.0 g). A vibrating ball-milling system was used to mix and grind all the prepared powder samples. The prepared samples were placed in the sample cup and the surface leveled off with a knife edge. The dispersion in calculated KM values by using this filling method was checked. Variations in ambient humidity and in packing density caused the values to vary between ± 3 % and ± 15 %. Each sample was therefore refilled several times, the KM values of each spectrum were calculated and their mean value was used to represent the sample. The same KBr reference spectrum was used in all the KM calculations for each sample.
Results
Calibration
Small amounts of pure strontianite were added to the strontianite contaminated BiSSCO powder diluted in KBr. Five standard samples with various content of strontianite were made from the same starting material i.e. BiSSCO diluted to 2.4267 wt % in KBr. The procedure for preparing the standard samples is described as follows: One basis sample of BiSSCO diluted in KBr (2.43 wt %, 4 g) was prepared by mixing and grinding for 10 min. and it was afterwards divided into five samples. Different amounts of pure SrCO3 (0.0265-0.0797 wt %) were added to each of these samples and they were ground and mixed separately for further 10 min. The spectra of the BiSSCO sample diluted in KBr with no strontianite added and the same mixture added 0.0797 wt % strontianite are shown in Figure 2. %.
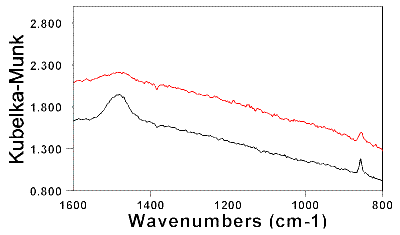
Figure 2. A BiSSCO sample spectrum with a carbonate content to be determined (at top) compared to a standard spectrum representing 0.0797 wt% strontianite added to a BiSSCO 2.4267 wt% sample.
In order to measure the carbonate peak height in the measured BiSSCO spectra a straight baseline was drawn between 1693.6 and 1361 cm-1 in the spectra, i.e. on both sides of the n 3 band shown in Figure 3. The drawn baseline follows in a reasonable way the steep slope of the absorbance curve observed in all the SPC spectra.
Figure 3. BiSSCO spectrum with the v3 fundamental strontianite spectral band.
Figure 4 shows the calculated differences in KM values at 1479.4 cm-1 between this baseline and the carbonate peak plotted as a function of strontianite concentration values. The straight line through the data is found by applying linear regression. The relationship between the KM- and concentrations values behaves linearly, it is therefore possible to extrapolate the regression line and calculate the amount of carbonate in the BiSSCO sample. The intercept of the concentration axis found by calculation is equal to 0.0383 wt %. The concentration of BiSSCO in the KBr matrix is 2.4267 wt %, which gives by calculation a total amount of carbonate in the BISSCO sample equal to 1.58 wt %(=100/2.4267* 0.0383 wt % =1.577 wt %).
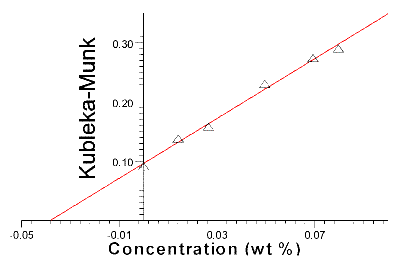
Figure 4. Kubelka-Munk values versus concentration values of SrCO3 added to diluted BiSSCO samples (method of standard additions).
Detection limit
An estimate of the detection limit of the two methods is found by comparing the measured spectra of a BiSSCO powder containing no carbonates and a BISSCO sample in which only small amounts of strontianite are added. Small amounts
(0.0023 g) of 0.0998 wt % strontianite diluted in KBr were added to a 3.10 wt % BiSSCO sample free of carbonates in KBr which was ground as described in the experimental section. This mixture was shaken vigorously (not ground) for 5 minutes. The strontianite concentration in the total sample is calculated to be 3.14* 10-4 wt % (» 3 ppm). This corresponds to a strontianite concentration in the analyzed BiSSCO sample of 0.01 wt % (=100/3.10* 3.14* 10-4 wt %). The spectrum of the BiSSCO sample free of carbonates was used as the reference spectrum and the KM-spectrum of the BiSSCO sample with added strontianite was recorded (100 scans were coadded for both samples). The calculated KM-spectrum showed a small absorption peak of the n 3 band of the carbonate ion. The detection limit might be improved by coadding more scans i.e. improving the signal to noise ratio. It can be debated whether the pure strontianite mixed and ground with KBr gives the same spectral response as comparable strontianite concentrations in the BiSSCO powders. The morphology of the strontianite and BISSCO powder particles might be different. The qualitative analysis revealed, however, that the spectral lines of the carbonates were observed at the same wavenumber positions indicating that no serious change of the crystal lattice has been made during grinding. In addition, the relationship shown in Figure 4 suggests that in this case it is acceptable to use pure substances in the calibration step.
Conclusions
Quantitative methods can be applied to measure the content of carbonates in the BiSSCO based powders. It was necessary to dilute the strongly absorbing powders in a non-absorbing potassium bromide matrix to concentrations less than 2.5 wt % in order to preserve the linear relationship between the KM- and concentration values. DRIFTS is found to be a very sensitive technique for detecting low concentrations of carbonates in BiSSCO like powders. Absorption peaks originating from strontianite added in low concentrations (» 3 ppm) to a BiSSCO powder free of carbonates were distinguished from the BiSSCO spectral features.
References
-
I.M. Hamadeh, S.A. Yeboah, K.A. Trumbull and P.R. Griffiths, Appl. Spec. vol. 38, 486, (1984).
-
T.L. Hughes, C.M. Methven, T.G.J. Jones, S.E. Pelham, P. Fletcher and C. Hall, Advn. Cem. Bas. Mat., 2, 91, (1995).
-
T.J. Porro and S.C. Pattacini, Appl. Spec., vol. 44, 1170, (1990).
-
A. Tsuge, Y. Uwamino, T. Ishizuka and K. Suzuki, Appl. Spec. vol. 45, 1377, (1991).
-
D.M. Hembree and H.R. Smyrl, Appl. Spec. vol. 43, 267, (1989).
-
J. Bak and B. Kindl, Appl. Spec. vol. 51, 1730, (1997).
-
J. M. Olinger and P. Griffiths, Appl. Spec. vol. 47, 687, (1993).
-
S. Agyare, S. Wang and P. R. Griffiths, Appl. Spec. vol. 38, 259, (1984).
-
K. Moradi, C. Depecker and J. Corset, Appl. Spec. vol. 48, 1491, (1994).
-
Z. Krivacsy and J. Hlavay, Spectrochimica Acta vol. 50A, 49, (1994).
-
G. Kortum, Reflectance spectroscopy. Springer, New York (1969).
-
W.WM. Wendlandt and H. G. Hecht, Reflectance Spectroscopy, in series of Chemical Analysis vol. 21, Interscience Publishers (1966).