Molecular Vibrations – a simple tutorial
3. Molecular Vibrations – a simple tutorial
Editor
Some years ago, I used to teach vibrational spectroscopy to our undergraduates in Chemistry. I found most of them were happy with the idea that molecular vibrations have certain characteristic frequencies when the sample contains contaminated specific chemical groups (so-called group frequencies) but they were very wary of ‘fundamental modes’. The group frequency approach is very popular amongst organic chemists and it is based on a good (if somewhat limited) theoretical basis. The problem is that users tend to stop at this level of sophistication and go no further. Eventually they get themselves into terrible trouble.
The group frequency approach and some beautiful animated examples are given in IR Tutor – available on CD-ROM from Perkin Elmer. It includes the out-of-phase deformation of toluene, which I always call the ” demented spider motion”. Get a glimpse of IR Tutor and you’ll see why!
My solution was to take our students through the vibrations of chloroform and then to assign the Raman spectrum. The project was written up and published in Spectrochimica Acta at the end of one of the series of special editions devoted to F-T Raman spectroscopy and it has been suggested a re-run brought more-up-to-date might be of value to the non-specialist readers of IJVS.
[PJ Hendra Spectrochemica Acta A51 (1995) 2205-2208]
Fundamental Modes
Chloroform, like all molecules perpetually vibrates (even at zero Kelvin) in a very complex manner. If you could see a molecule under a hyper microscope you would note that it was contorting incredibly rapidly, rotating much more slowly only around 20 billion rpm and moving through space (at a speed of the order of 1000km/hr).
The linear movement is called translation by spectroscopists and the rotational motion rotation (even we spectroscopist’s ability to invent jargon runs into the sands!) We are not concerned here with either – only the molecular vibrations.
If you wished to exactly define the shape and size of a molecule, where it is placed and its orientation, you could do it by stating a set of cartesian co-ordinates for each atom’s nucleus referenced to axes in your instruments’ sample area. Let’s draw up HCl (I’m too lazy to trot out all this rubbish for chloroform).
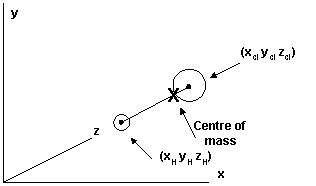
The position of the centre of mass of the molecule within the instrument is all you need to know to define where the molecule is [we will ignore its orientation]. The orientation must be defined around the centre of mass so whatever else we say about the vibrations one point is clear, the total number of cartesian co-ordinates must restrict the vibrations and their complexity.
Put it this way – in chloroform, we have 5 atoms so the number of XYZ co-ordinates we have is – 3 x 5 = 15 —- but 3 simply tell us where the wretched molecule is!
Now 3 are ‘burned up’ telling us its orientation. So we have 15-6 possible ways of defining the vibrations at most.
The important things to remember about vibrations is that they have properties which are quite fundamental. They are
1. All the atoms move periodically and in phase
2. the centre of mass remains fixed at all times and
3. the molecule retains its orientation and position throughout the motion.
So – returning to dear old HCl we get
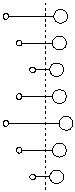
Note that the big fat chlorine hardly moves. The sprightly hydrogen belts back and forward like a table tennis ball. This is because the centre of mass must remain fixed.
Now Chloroform – the complex vibrational motion is made of a series of simpler fundamental vibrational motions – and there can’t be more than nine of them.
Draw on a piece of paper five or six structures of chloroform. Remembering the rules above try to draw the vibrations using vectors – you won’t get them all – try to draw three. To give you the idea I will do the job for you for H2O
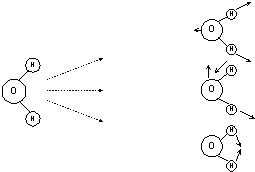
(This is shown in action in IR tutor)
So have a go at chloroform and see how well you can do.
STOP HERE

