Cosmetic Spectroscopy: A review of infrared and Raman studies of skin and hair
4. Infrared and Raman Studies of Skin and Hair:
A review of cosmetic spectroscopy
Kathleen Martin
Unilever HPC USA
3100 Golf Road
Rolling Meadows, IL 60008
USA
Tel: (847) 734-3714
Fax: (847) 734-3686
email: kathleen.martin [at] unilever.com
Introduction
Each year the cosmetic industry reaps billions of dollars from sales of products designed to enhance skin and hair. Products to cleanse, moisturize or condition skin and hair abound. In order to compete for the consumer’s dollar, the industry has expended much effort on improving our understanding of the composition and structure of skin and hair and on the nature of the interactions between these substrates and the products themselves. Vibrational spectroscopy, both in vitro and in vivo, has been an important tool in these studies. Presented here is an overview of infrared, Raman and near-infrared spectroscopic studies of skin and hair in the cosmetic industry.
- Composition and Structure of Skin and Hair
Skin and hair both function as protective barriers between the body and the environment. The outermost layer of skin, the stratum corneum (SC), is of particular interest to cosmetic scientists as this is the layer affected by cosmetic products. In addition to defending the body against chemical and bacterial attack, the SC regulates evaporative water loss through the skin. Scalp hair plays a similar role by acting as a cushion for the head, protecting the scalp from sunlight, and helping to maintain body heat. Both skin and hair are composed primarily of keratin, a tough, insoluble protein also found in nails, hooves, horns and feathers, plus smaller amounts of lipids and water. The similarities and differences are illustrated in Figures 1 and 2 which compare the mid- and near-infrared spectra of skin and hair. In the mid-infrared, strong amide I and II bands from keratin protein occur around 1660 and 1550 cm-1for both skin and hair. Differences are seen in the intensities of the C-H stretching region (due primarily to lipid alkyl chains) and in the region below 1400 cm-1. In the near-infrared, the skin spectrum is dominated by the strong water bands at 1920 and 1440 nm, while the hair spectrum has strong protein bands at 1500, 2050 and 2180 nm in addition to the water bands, indicative of differences in the relative amounts of water and protein in the two tissues.
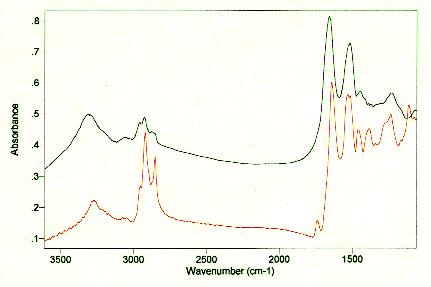
Figure 1. Mid-infrared spectra of hair (top) and skin (bottom). The hair spectrum was acquired by diffuse reflectance on short pieces of hair mixed with KBr. The skin spectrum was acquired on a forearm in vivo by attenuated total reflectance using a 40° ZnSe internal reflectance element. (Spectra collected on a Bio-Rad FTS 40 FTIR.)
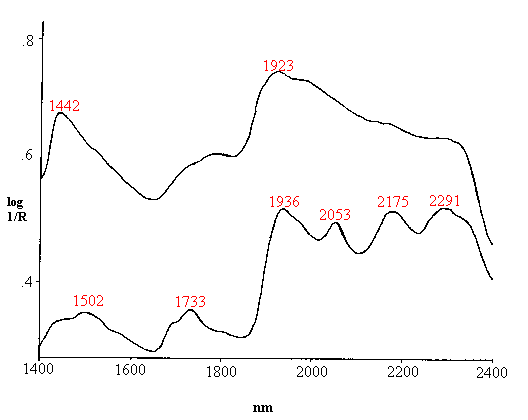
Figure 2. Near-infrared spectra of skin (top) and hair (bottom). The spectra were acquired via a fibre optic cable on a grating spectrometer (LT Industries Quantum 1200).
Although the functions and compositions of the SC and hair are similar, the structures of the two are very different. The SC is a stratified structure about 10-40 m m in thickness, with significant variability in composition between the deeper and surface layers. At the base of the SC, living cells extrude lipids and cornify into flattened, anucleate cells which are pushed outward until they are shed upon reaching the skin surface. Below the SC are the living epidermis, about 100-200 m m thick, and the dermis, about 2-4 mm thick (Figure 3).
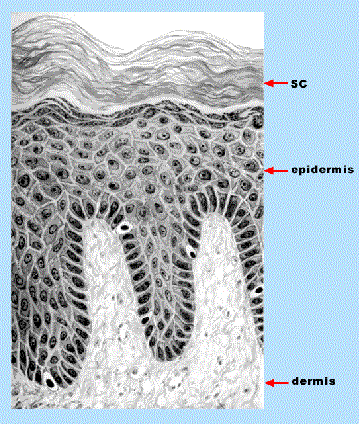
Figure 3. Diagram of a skin cross-section showing the multi-layered stratum corneum (SC), living epidermis and dermis. Adapted from D. M. Pillsbury, W. B. Shelley and A. M. Kligman, “Dermatology”, W, B. Saunders Co., Philadelphia, 1956
Hair, on the other hand, is composed of a cylindrical cortex about 40-100 m m in diameter surrounded by an outer protective sheath of overlapping cuticle cells arranged like shingles on a roof. New cuticle at the base of the hair shaft consists of about 10 cell layers, altogether about 5m m thick. The cuticle cells gradually erode away over time as hair is increasingly exposed to UV radiation, detergents, and combing so that tip end hair may have few protective cell layers in the cuticle. Figure 4 shows SEM photomicrographs of new growth and weathered hair. The latter shows cuticle uplift, chipping and cracking, which result in a dull appearance of the hair and contribute to a rougher feel.

Figure 4. SEM photomicrographs of new growth (top) and tip end (bottom) hair. Longer exposure to sunlight, wind and grooming causes cuticle uplift, chipping and cracking.
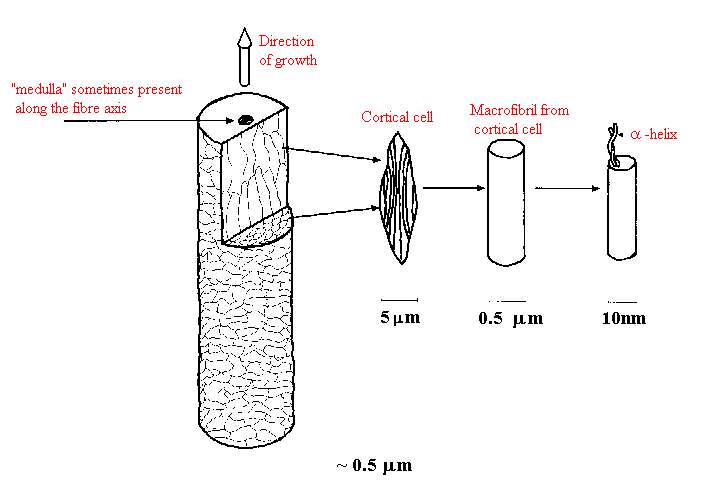
Figure 5 illustrates the substructure of hair.
The tightly packed cortical cells, in turn, are composed of crystalline keratin, mostly in an a -helical arrangement, plus less organized protein structures. Figure 5 illustrates the substructure of hair.
Lipids are an integral part of the membrane structures that act as a glue to hold the cuticle cells onto the cortex in hair. These lipids are primarily free fatty acids and cholesterol, but cannot be distinguished analytically from the sebaceous lipids which are always present. Sebum, in particular, affects foaming properties of shampoos, as well as oiliness and frictional behavior of the hair. In skin, lipid composition and structure are important in maintaining skin suppleness and elasticity and are also thought to be responsible for both the barrier to evaporative water loss and the water holding capacity of the SC.
Water content in both skin and hair depends strongly on the relative humidity. Hair, in fact, acts as a hygrometer. Below 70% relative humidity (RH) water content in hair and SC is quite similar, about 10-30% by weight [1]. Measurements of skin tissue by electron probe analysis have indicated that water content within the SC gradually increases from about 10% to about 30% between the surface and deeper layers, followed by an abrupt increase in water content below the SC to about 70% water [2].
Changes in the water or lipid content of skin or hair may affect the feel or look of hair and skin by altering perceptions of dryness, elasticity or brittleness. Many of the products currently on the market or in development are designed to combat these problems and thus many of the spectroscopic measurements are geared toward evaluating these components.
Spectroscopic Approaches to Skin and Hair Measurement
- Attenuated Total Reflectance on Skin
The desire to measure skinin vivogenerally limits the number of approaches available for collecting spectroscopic data. ATR (attenuated total reflectance) has been applied extensively for infrared spectroscopy skin measurements because of its ease and rapidity of use. In ATR, a sample is placed on an infrared transparent crystal of high refractive index (the internal reflectance element or IRE), the geometry of which permits almost total internal reflection. The internal reflectance results in an evanescent wave which is attenuated in regions where the sample absorbs the radiation. The depth of penetration of radiation is quite shallow. For skin, it is calculated to be on the order of 1 micron in the mid-infrared, ideal for measurements of cosmetic treatment effects, where only the skin surface is of interest.
ATR was initially proposed by Scheuplein as an alternative to reflection measurements on skin where scattering can be a problem [3]. Ferren later used ATR with a dispersive infrared instrument to measure products deposited on skin or hair [4]. He used untreated skin as the reference, so that he could look at product in situ, without interference from skin absorption bands. Photos show the author with his head directly on an ATR plate in order to measure a hair lotion and with his cheek pressed against an ATR plate to measure an aftershave lotion. The first ATR study to examine skin itself was performed by Puttnam and Baxter, using a V-shaped KRS-5 (a mixed crystal of thallium bromide iodide) ATR crystal to obtain two reflections off of the fleshy part of the hand [5]. They found only minor variations between the ATR spectra and transmission spectra obtained from thin slices of skin. The greatest variability between individuals was seen for the water band intensity around 3400 cm-1, attributed to perspiration effects. Concern about the toxicity of KRS-5 caused a switch to Irtran II (ZnS) [6]. The ATR technique was later improved by the use of a horizontal ATR crystal, making it possible for subjects to simply rest their arm on the IRE during data collection [7] (Figure 6).

Figure 6. Photo of subject with her arm resting on a horizontal ATR accessory designed specifically for skin studies (Spectra-Tech).
In addition, the degree of contact between the skin and the IRE affects the spectrum. Baier found that application of creams or oils resulted in stronger amide I and II absorbances as contact between skin and IRE improved [7]. Klimisch and Chandra handled this problem by holding a damp towel to the skin before each measurement, ensuring reproducible contact [8]. In this case, they were looking at the substantivity of polydimethylsiloxanes on the skin rather than at hydration, so their approach was suitable.A drawback to ATR is that the depth of penetration of radiation into the skin is variable. The depth depends on the wavelength, the angle of incidence, and the refractive indices of the IRE and the skin. Treatment of the skin with lotions or hydration of the skin through increased relative humidity generally decreases skin’s refractive index, thus decreasing the depth of penetration of radiation. Hydration can also be induced by occlusion, such as that between skin and the IRE during the acquisition of an ATR spectrum. Because of the varied nature of the SC at different depths, spectra acquired before and after a treatment that affects the skin refractive index are not always comparable.
A common practice in skin studies is tape-stripping, where successive layers of skin are removed by scotch tape, followed by ATR measurements of the freshly exposed skin surface. Removal of layers of SC results in an increasingly hydrated surface, reducing skin’s refractive index and generally increasing the degree of contact between skin and IRE. Despite the uncertainties due to variability in refractive index and in degree of contact, ATR remains a valuable method of skin measurement as it is rapid and extremely simple to use.
- Raman Spectroscopy on Skin
Raman spectroscopy, with its relative insensitivity to hydration state of the SC, is increasingly popular for the study of skin. Raman spectra give information about protein conformation, lipid structures, disulfide bonds and amino acid side chain groups. The complementary nature of IR and Raman is illustrated by Barry, et al. who give assignments for transmission infrared and Raman scattering spectra of excised skin [9]. Assignments were also given in a later study by the same group comparing FT-Raman spectra of excised human stratum corneum, callus removed from the bottom of feet, plus hair and nails [10]. Although the spectra of the four tissue types were overall quite similar, some striking differences were noted. Stratum corneum tissue had a strong band around 2883 cm-1, attributed to methylene groups in lipid chains, which was mostly absent in the spectra of the other tissues. In addition, the hard keratins in hair and nails clearly showed the presence of more disulfide groups (510-530 cm-1) than appeared in the soft keratins of SC and callus.
Raman data obtained in vivo and in vitro were compared using normal Raman scattering, a Raman microprobe and FT-Raman using near-infrared excitation [11]. The latter configuration was used to examine forefingers before and after tape-stripping. The most striking difference between the in vivo and in vitrospectra was the presence in the former only of a strong band near 3230 cm-1, believed to be due to N-H stretching. In addition, the in vivo spectra showed more intense C-C lipid skeletal backbone stretching vibrations between 1030 and 1130 cm-1. A comparison of the three Raman techniques indicate that significant differences occur in relative band intensities, probably due to orientation effects, but no differences in band frequencies.
Raman spectroscopy using a confocal microscope represents a recent approach that holds a lot of promise for in vivo skin studies since the confocal microscope allows depth-profiling to be performed. In a recent paper, the use of a confocal Raman on different anatomical sites was described [12]. Spectra acquired at the skin surface indicated significant regional variation in SC composition such as between fingertips and forearms.
- Near-Infrared
Near-infrared diffuse reflectance spectroscopy has also been applied to skin, although this approach suffers from a poorly understood depth of penetration of the reflectance signal. Scheuplein discussed the optical pathways through skin from the UV to the infrared, and how the stratification of the SC enhances the possibilities of internal reflections [3]. Figure 7 shows possible pathways for light to interact with skin.
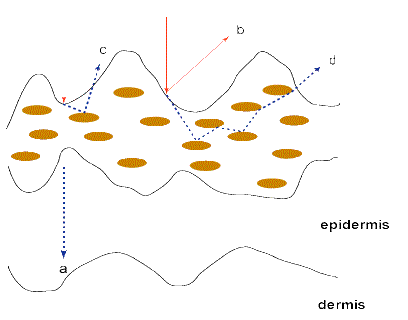
Figure 7. Optical pathways for light in skin: a) absorbance or transmission, b) regular reflectance, c) diffuse reflectance, and d) multiple internal scattering.
Scheuplein assigned a value of 1.55 to the refractive index of the SC, and calculated a minimum of 4% regular (surface) reflectance based on the Fresnel equations. The off-normal angles of incidence produced by the irregular SC surface are expected to make the amount of regular reflectance higher [13]. Scattering from the skin surface is not appreciable below about 2000 cm-1, but is still significant in the near-infrared region and has been applied to studies of skin moisturization [14]. The lack of specificity of information regarding lipids and protein and uncertainty in the depth of penetration of radiation make this approach generally limited to information about water content and surface texture [15].
- Hair
Hair, in contrast, is generally much easier to sample than skin as it is essentially in the same state whether it is attached to the scalp or not. Unless one is looking at the hair follicle, hair detached from a head differs only in that continual exposure to sebum is absent [See Section 1]. Thus, many spectroscopic approaches are available for examining hair. These include transmission, ATR, photoacoustic spectroscopy (PAS), infrared microscopy in the mid-infrared, Raman scattering and Raman microscopy, and near-infrared diffuse reflectance.
A comparison of infrared microscopy, diffuse reflectance and ATR approaches for examining oxidative hair damage was made by Joy and Lewis [16]. ATR/KRS-5 was found to be the least reproducible method while diffuse reflectance spectra of fibres mixed with KBr produced very noisy spectra. Transmission spectra of flattened fibres allowed one to sample specific locations in a fibre and yielded better spectra. In another study, infrared spectra obtained using ATR with either a ZnSe or diamond IRE were compared to spectra from transmission microscopy in an examination of oxidative effects in hair fibres [17]. These authors also found the microscopy approach to yield good reproducible spectra. ATR/ZnS was found to be too irreproducible to be of value, although ATR/diamond, using 7-9 fibres at a time, was more succcessful.
Although infrared microscopy is a popular approach for sampling hair fibres, it generally does not allow for high spatial resolution. The use of synchrotron radiation to achieve a spatial resolution of a few micrometers with infrared microscopy was described Bantignies, et al. [18]. The use of a high intensity source resulted in the ability to map across a fiber cross-section to study ingredient penetration into a fibre or oxidation effects at different depths.
Confocal Raman microscopy offers another means to achieve high resolution, this time in three dimensions [19]. FT-Raman has also been applied to studies of hair and assignments for the Raman bands of hair are given in references [20] and [21]. Raman spectroscopy, even with a near-infrared laser source, is generally limited to unpigmented hair due to enormous fluorescence from melanin.
Near-infrared reflectance spectroscopy allows bundles of hair rather than single fibres to be sampled and can be used in situ with a fibre optic probe. Ozaki, et al. used near-infrared reflectance to study water content in hair [22].
When data from large numbers of individuals are desired, spectroscopic measurements may be performed away from the laboratory. The advent of more portable instruments makes it possible to collect spectra in unexpected settings. Figure 8 shows the collection of near-infrared data in a salon.

Figure 8. Photo of near-infrared spectra being acqiured in a beauty salon. The spectrometer is a grating instrument from LT Industries, with a fibre optic cable.
Applications
The main objectives of spectroscopic studies are generally different for skin and hair, although some overlap occurs. Skin studies are concentrated on product deposition and penetration, measurements of skin “dryness”, plus an understanding of protein and lipid conformation. Most of the published studies on hair, on the other hand, concern oxidative effects of chemicals or sunlight. Below, the major areas of application for the spectroscopy of skin and hair in the cosmetic industry are discussed.
- Measurement of Ingredients Applied to Skin and Hair
As mentioned earlier, Ferren was among the first to use infrared spectroscopy to make measurements of living skin [4]. He demonstrated the ability to measure a variety of products deposited on the skin, including deodorant, hand lotion and an after-shave, by ATR. Also included were measurements of a hair preparation after application to the hair, requiring the subject to place his head onto the ATR crystal.
Many of the other early infrared in vivo studies also focused on measurement of products deposited on skin. The substantivity of a skin cream on skin was demonstrated by Puttnam and Baxter, who noted increased infrared intensities of methylene stretching and deformation modes from the presence of fatty alcohols and mineral oil plus a weak feature at 1720 cm-1 due to stearic acid [5]. After washing, traces of the skin cream remained. Puttnam later studied the uptake of sodium dodecylbenzene sulfonate onto the palm of the hand [6]. Uptake was found to increase with either increasing surfactant concentration or with increasing time of exposure, but was decreased in the presence of an ether sulfate, presumably due to competition for sites on the skin.
Somewhat more recently, ATR was used to look at the deposition and wash-off of siloxane polymers of differing molecular weights from skin [8]. Siloxane polymers are commonly used in skin care products because they impart a smooth, non-oily feel, and are non-toxic and non-irritating. The siloxane polymers are ideal for infrared analysis because they have a distinctive absorption band at 1261 cm-1. The amide II band of skin protein (at about 1550 cm-1) was used as an internal reference for degree of contact with the ATR crystal as it is not influenced by water content the way the amide I band (at about 1660 cm-1) is due to overlap with the water deformation band. A calibration curve was constructed by depositing known amounts of siloxane polymer onto the skin via a volatile solvent. After three soap and water washings, about 38% of the high molecular polymer was found to remain on the skin, while only about 9% of the lowest molecular weight polymer remained.
The same group developed a different infrared method to determine the amount of siloxane polymers deposited on hair from a conditioner [23]. Diffuse reflectance was proposed as a simple and quantitative method, with the degree of dilution in KBr and grinding time optimized. A calibration curve was made using atomic absorption spectroscopic measurements, and a linear relationship between the two methods was found for siloxane polymer concentrations of 250 to 1840 ppm on hair.
A more subtle approach to studying ingredient interaction with the skin was described by Dreassi, et al. [24]. They used near-infrared reflectance in vivo to look at changes in normal and atopic (very dry) skin after application of different ingredients. Treatment resulted in differences in spectral absorption, depending both on the nature of the ingredient and on the skin classification. Principal components analysis (PCA) was used to distinguish the responses of the normal and atopic groups. Differences were explained as due to differences in water distribution between the surface and deeper layers and by different interactions between water and lipids.
In another near-infrared reflectance study, application of moisturizing lotions to the skin resulted in decreased scattering effects in the skin spectra but no change in the SC water band intensities [25]. It was concluded that the lotions acted to smooth the skin surface, rather than to hydrate it .
- Water in Skin and Hair
A major cosmetic concern is dry skin. This condition can be brought about by low humidity, excessive exposure to detergents, UV radiation, or by aging. It is often assumed that dry skin is caused by a lack of moisture and thus measurement of water is of great interest in assessing dry skin and in determining whether adding water is an effective means to combat dry skin.
Gloor and co-workers used ATR extensively to monitor changes in skin water content after removal of surface skin lipids [26], and as a function of sex and age and after application of O/W and W/O emulsions [27]. Water content was measured as a “moisture factor” (MF), which consisted of the ratio of intensities of the amide I and II bands. The ratio increases upon hydration due to the contribution of the water deformation mode to the measured amide I intensity. Skin was found to be hydrated by the application of skin creams and by surfactant and soap solutions. No significant differences in MF were found due to sex or age.
A thorough review of the use of ATR infrared spectroscopy to measure water in skin was given by Potts [28]. In it, he criticized the use of the amide I/II ratio as a measure of water content because, although the amide I band increases in intensity with increasing water content, other changes to the protein that occur upon hydration may also affect the spectra. Potts’ laboratory approached the determination of skin water content using the 2100 cm-1 combination band of water [29]. In order to correct for the degree of contact with the ATR crystal, the integrated area of the 2100 cm-1 peak was normalized to the area under the baseline to correct for scattering. The advantage of using this band is its lack of interferences from skin or ingredient components. In vitro data showed a good correlation between the absorbance at 2100 cm-1 and the relative humidity. Potts’ method was later applied by Edwardson, et al. to study water content in skin after occlusion by dressings with varying water uptake properties [30].
The use of near-infrared reflectance to measure water in skin was first proposed by Walling and Dabney [14]. The kinetics of water desorption from porcine skin were measured on samples treated with water, glycerol and a glycerol/mineral oil-based skin lotion. The NIR spectra indicated that water loss from the skin was retarded by the lotion, but not from glycerol alone. In the same paper, in vivo NIR measurements were found to correlate to clinical subjective ratings of dryness. In a study of dry skin by deRigal, et al. near-infrared measurements correlated better to clinical scores of dryness than did electrical conductance measurements, a traditional method for evaluating skin moisturizers [31]. In the same study, isolated stratum corneum tissue was analyzed by differential scanning calorimetry for water content which was was found to correlate highly with the difference in near-infrared absorbance at 1936 and 1100 nm.
A slightly different approach to the assessment of dry skin by NIR was taken by Dreassi, et al. [32]. Differences between subjects with normal and atopic (very dry) skin were studied by near-infrared spectroscopy using principal components analysis (PCA) to better resolve differences between the groups. Although no discrimination was able to be made between normal and atopic skin, interpretation of the PCA loadings suggested that normal subjects varied more in lipid structures, while atopic subjects varied more in what was termed “water equilibrium”.
Near-infrared reflectance spectroscopy was also successfully used to probe the hydration state of fingernails using the water combination band near 1980 nm [33]. Shorter wavelengths were not useful because they were found to probe so deeply into the tissue that reflectance bands corresponding to oxy- and deoxyhemoglobin were seen.
Differentiation between types of water in skin, based on hydrogen-bonding differences, was observed in the second derivatives of near-infrared reflectance spectra [15]. Water bands associated largely with lipid bilayers (1880 nm), primary and secondary water of hydration on protein (1928 and 1910 nm, respectively), and free water (1890 nm) were assigned. Differences in the distribution of water types were seen for different anatomical sites due to differences in SC thickness with consequently different depths of penetration of near-infrared radiation. Figure 9 compares the 2nd derivative spectra of water and skin in the water combination region.
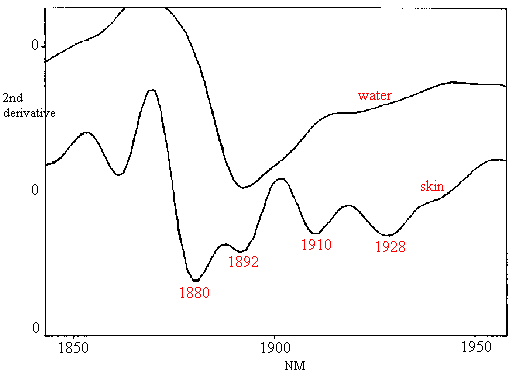
Figure 9. Second derivative near-infrared spectra of water (top) and skin in vivo (bottom).
The hydration state of human hair has also been studied using near-infrared reflectance. Ozaki, et al. used FT-NIR to measure water content in hair fibre bundles [22]. After normalization to the second derivative intensity of protein at 2058 nm, the second derivative intensity of water at 1925 nm was found to correlate to independent moisture measurements based on changes in dielectric constant. The dependence of water content in hair on relative humidity (RH) is illustrated in Figure 10, which shows near-infrared spectra of hair at different RH. The intensities of the 1940 nm combination band and the 1440 nm first overtone of water decrease dramatically as the humidity increases. Figure 11 is a plot of the 2nd derivative water band intensities at 1913 and 1931 nm (primary and secondary water of hydration) as RH first decreases, then increases. The hysteresis seen between the drying and humidifying cycles was earlier measured by gravimetry [34].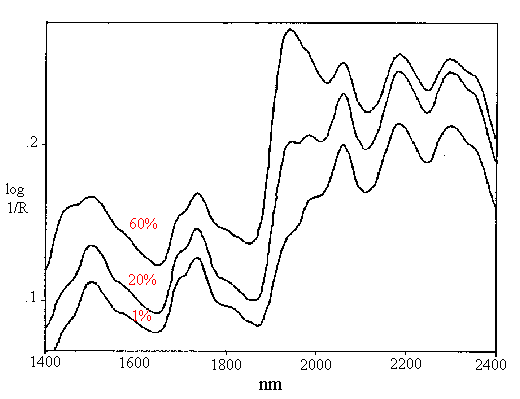 Figure 10. Near-infrared spectra of hair equilibrated at 60%, 20% and 1% relative humidity.
Figure 10. Near-infrared spectra of hair equilibrated at 60%, 20% and 1% relative humidity.
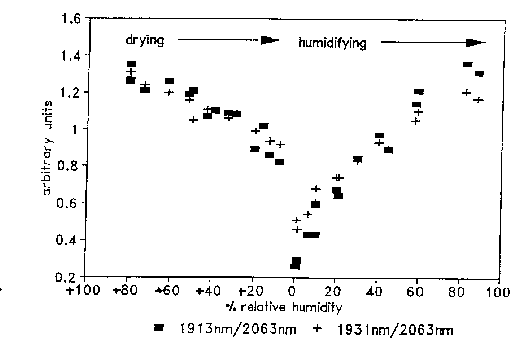
Figure 11. Plot of second derivative near-infrared intensities for primary (1913 nm) and secondary (1931 nm) water of hydration on protein, normalized to the intensity of the protein band at 2063 nm.
- Skin Lipids
The conformation of the SC lipid bilayers is thought to be important to their ability to maintain the skin barrier function, as well as to maintaining a desirable skin feel and plasticity [35].
A correlation between SC lipid structure and water flux through the SC was suggested by Golden, et al. based on changes in infrared spectra [36]. As excised porcine SC membranes were heated from 30 – 115° C, the spectral bands due to the antisymmetric and symmetric stretches of the lipid alkyl chain methylene groups broadened and shifted to higher wavenumber. The shift occurs because of an increase in the number of gauche conformers (increased disorder). At the same time, an abrupt increase in water permeability occured near 70° C, where the increase in the shift of v CH2 was greatest.
In vivo studies of SC intercellular lipids were conducted on progressively tape-stripped forearms by ATR-infrared spectroscopy [37]. Disorder, as gauged by an increase in wavenumber of the methylene stretching vibrations, decreased through the first four tape strips, each tape strip roughly corresponding to one cell layer. Beyond the first four tape strips, the degree of disorder remained constant.
The degree of conformational order/disorder in SC lipids was also studied by Denda, et al. using ATR to look at shifts in the C-H stretching frequencies in normal and surfactant-induced scaly skin [38]. A correlation between increased wavenumber and increased transepidermal water loss was found for normal skin, but not for scaly skin, where other mechanisms were expected to contribute to barrier function.
Raman spectroscopy has been used to probe lipid structures in skin in vitro. Differences in lipid composition were found between the different layers of skin and between subjects with the presence of ceramide III identified through features at 1061, 1128 and 1296 cm-1 [12]. Raman spectroscopy was also suggested as a means for obtaining useful information on the nature of SC lipid bilayer packing in the hydrocarbon chain region (1000-1150 cm-1) [9].
- Proteins in Skin and Hair
Microtomed horse hair was studied by Bendit using infrared transmission and assignments were made for a -, b -, and non-crystalline amide bands [39]. Polarization measurements were employed to distinguish crystalline and non-crystalline phases. Hydration of the hair resulted in shifts of the amide A, I and II frequencies for the non-crystalline phase, indicating the formation of water bridges and consistent with the high swelling capability of this secondary structure. Much smaller shifts in the amide I and II bands, and no shift in the amide A band, were seen for the two crystalline phases, indicating that hydrogen bonding strength was not affected by hydration. Rather, the spectra indicated that a weak interaction between water molecules and the carbonyl of the peptide group exists. Difference spectra indicate that the hydrogen bonds formed between water and peptide groups are stronger than those formed between peptide groups, while both of these bonds are weaker than water-water hydrogen bonds.
ATR was used to look at human hair by Baddiel [40]. Using a refractive index for hair of 1.55, the depth of penetration into the hair was calculated to be about 3.5 µm and thus the majority of the signal originated from the cuticle. Measurements were performed on intact hair and on hair with the cuticle removed. The cuticle was concluded to be composed primarily of a -keratin, with a significant amount of
b -keratin present, while the cortex was found to consist of a -keratin plus random coil or amorphous forms.
FT-IR photoacoustic spectroscopy was used to distinguish cuticle and cortical regions of human hair [41]. The thermal diffusion length calculated for hair indicated a depth resolution of about 1 µm. As the optical velocity of the mirror decreased and sampling depth increased, shifts occured for both the amide I and II bands due to changes in protein composition. A relatively large frequency shift was found to correspond to the transition from the cuticle to the cortex, allowing an estimate of cuticle thickness to be made.
Confocal Raman was also used to depth-profile human hair [19]. Differences between cuticle and cortical proteins were evident. The amide III band at 1246 cm-1 was used as a marker for disordered protein along with bands at 1338 and 1313 cm-1. The occurrence of the amide I band at 1656 cm-1 was concluded to be consistent with a predominantly a -helical secondary structure. In a separate study, the frequencies of the amide I and III bands were found at 1652 and 1274 cm-1, respectively, in Raman spectra of excised human SC, taken to be indicative of a predominantly a -keratin structure [9].
ATR spectra of human nails indicated that the outermost layer of nail contains a significant fraction of a -helical secondary structure, and a lesser degree of b -sheet structure [33]. The intermediate layer, on the other hand, was suggested to have few or no a -helices and is thus mostly a disordered structure. The deepest layer of the nail is very thin (about 2 cell layers), but appeared to be similar in structure to the dorsal (outermost) layer.
- Oxidation of Hair
Hair keratin undergoes oxidation by both photochemical (exposure to UV radiation) and chemical means (bleaches, permanent waves or permanent dyes). Oxidation results in decreased tensile strength of hair due to disulfide bond scission, in color changes due to melanin degradation, and in more easily abraded cuticle.
The products of the oxidation of the disulfide group of cystine, in particular, are conveniently monitored by infrared spectroscopy. Hair fibres, with diameters of 60-100 m m, are too thick for transmission spectroscopy without first thinning the fibres in a press. Various other infrared sampling approaches have been taken, including ATR [16, 17, 42, 43], KBr pellets [44], diffuse reflectance [16], a diamond anvil cell [45], IR microscopy [16, 17] and synchrotron radiation IR microscopy [18].
The usual products from disulfide oxidation are the sulfonic acid of cystine (cysteic acid), cystine monoxide (R-SOS-R) and cystine dioxide (R-SO2S-R), with characteristic infrared absorptions at 1040, 1075 and 1125 cm-1, respectively. Comparison of root end and tip end hair allows one to examine the effects of natural weathering. Cysteic acid was found to increase going from root to tip, although the degree of increase was found to be quite variable (16, 17, 44). Figure 12 shows the infrared spectra of a single hair fibre at various distances from the root. As weathering (exposure to sunlight, wind, grooming, etc.) increases, the intensity of the cysteic acid band at 1040 cm-1 increases. Peroxide bleaching also produced increased cysteic acid content [16, 17], while treatment with meta-bisulfite to perm the hair resulted in an increase in cysteine-S-thiosulfate (Bunte salt), seen at 1022 cm-1 [16, 45].
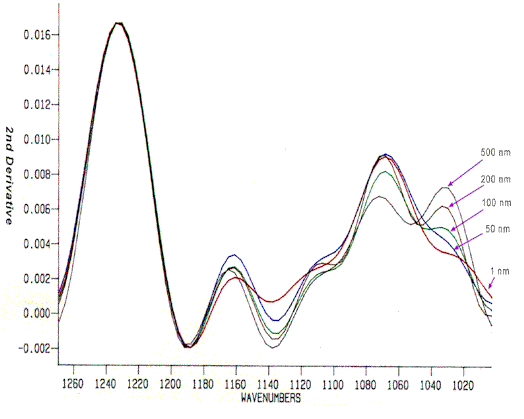
Figure 12. Transmission infrared spectra obtained on a single hair fibre at varying distance from the root (spectra acquired on a Bio-Rad FTS 45 FTIR).
Cross-sections of bleached and normal hair were compared using infrared microspectroscopy with synchrotron radiation [18]. Mapping of the 1040 cm-1cysteic acid band indicated a heterogeneous distribution through the cortex. A new band, at 1572 cm-1, was seen in the medulla, suggesting that bleaching produces some modification to the core of the fibre. Protein NH and lipid CH distributions were not affected by bleaching.
The role of relative humidity on disulfide oxidation was studied under artificial weathering conditions, using an Atlas weather-o-meter [43]. Oxidation within the cuticle, measured by ATR, increased with increasing exposure to radiation, but was not affected by humidity level. This was in contrast to wet mechanical properties, primarily a function of the cortex, which were most affected at high or low humidities, and least at intermediate humidities.
Artificial irradiation was also employed by Hoting, et al. to examine the effects of irradiation of hair by different portions of the spectrum on hair fibre properties [42]. Disulfide oxidation within the cuticle was found to be the same for light and dark-colored hair, in contrast to bulk fibre properties, which are degraded to a greater extent in light-colored hair due to the relative lack of melanin photo-protection. Visible and UV-A radiation were found to result in the highest cysteic acid levels.
Raman spectroscopy has also been employed recently to look at oxidation effects in hair. FT-Raman spectra of unpigmented hair were obtained by Pande who monitored the decrease in the disulfide band intensity at 510 cm-1 upon weathering, bleaching or reduction with a thioglycolate [20]. Cysteic acid, at 1045 cm-1, increased with bleaching, while S-H groups, at 2568 cm-1, increased upon weathering or reduction of the hair. Akhtar, et al. also used FT-Raman to study hair fibres before and after bleaching [21]. In addition to changes in the disulfide and cysteic acid band intensities, they found decreased tyrosine in bleached hair (853 and 825 cm-1) plus changes in the relative intensities in the amide I and III bands, the methylene bending modes and deformation modes 1300-1460 cm-1. The appearance of a new shoulder at 1652 cm-1 was assigned to the presence of b-sheet structures. Figure 13 compares the FT-Raman spectra of bleached and unbleached blonde hair. Bleaching results in a large increase in cysteic acid at 1040 cm-1 and changes in the region 500-450 cm-1, corresponding to disulfide bond breakage.
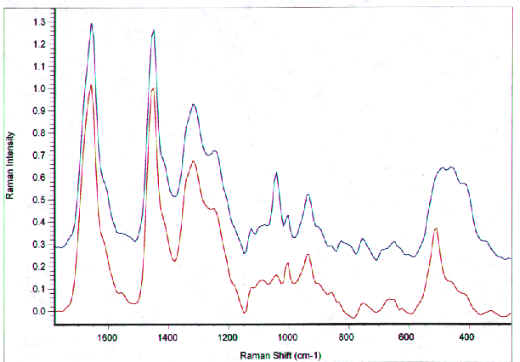
Figure 13. FT-Raman spectra of virgin and bleached unpigmented hair tresses (spectra acquired on a Nicolet 950 FT-Raman).
Conclusion
Infrared, near-infrared and Raman spectroscopies have proved to be valuable methods for studying a broad set of structural and functional properties of skin and hair. In many cases, these spectroscopies can be applied in vivo or in situ to give direct information about the state of skin or hair before and after treatment with cosmetic products.
The types of applications have evolved from being primarily concerned with the deposition of material onto skin or with the oxidative state of hair to studies of tissue hydration, lipid and protein conformation, and even of the physical state of the surface using scattering effects. The advent of depth-profiling using techniques such as confocal Raman microscopy and new mapping and imaging capabilities in infrared, near-infrared and Raman instruments are likely to be of great importance in future studies of skin and hair, allowing greater structural detail to be revealed. Instrument miniaturization is another important trend in spectroscopy which should affect cosmetic chemists by allowing increased portability, and thus the ability to acquire data on consumers in a variety of settings.
Armed with information about the deposition of materials onto skin or hair, changes in water content or lipid state upon application of products, or the prevention of deleterious effects from the weather, cosmetic chemists can provide guidance for product development or help to establish product claims.
Acknowledgements
The author wishes to thank Antonio Lopez for his assistance with the figures and Tiffany Lucas for the SEM micrographs.
References
- J-L. Leveque, “Water-keratin interactions”, in Bioengineering of the Skin: Water and the Stratum Corneum, P. Elsner, E. Berardesca and H. I. Maibach, Eds., CRC Press, Boca Raton, 1994.
- R. R. Warner, M. C. Myers and D. A. Taylor, “Electron probe analysis of human skin: Determination of the water concentration profile”, J. Invest. Dermatol. 90, 218-224 (1988).
- R. J. Scheuplein, “A survey of some fundamental aspects of the absorption and reflection of light by tissue”, J. Soc. Cosmet. Chem. 15, 111-122 (1964).
- W. P. Ferren, “MIR infrared spectroscopy in skin analysis”, Am. Perfumer and Cosmetics 84 (12), 27-36 (1969).
- N. A. Puttnam and B. H. Baxter, “Spectroscopic studies of skin in situ by attenuated total reflectance”, JSCC 18, 469-472 (1967).
- N. A. Puttnam, “Attenuated total reflectance studies of the skin”, JSCC 23, 209-226 (1972).
- R. E. Baier, “Noninvasive, rapid characterization of human skin chemistry in situ”, JSCC 29, 283-306 (1978).
- H. M. Klimisch and G. Chandra, “Use of Fourier transform infrared spectroscopy with attenuated total reflectance for in vivo quantitation of polydimethylsiloxanes on human skin”, J. Soc. Cosmet. Chem. 37, 73-87 (1986).
- B. W. Barry, H. G. M. Edwards and A. C. Williams, “Fourier transform Raman and infrared vibrational study of human skin: Assignment of spectral bands”, J. Raman Spectrosc. 23, 641-645 (1992).
- A. C. Williams, H. G. M. Edwards and B. W. Barry, “Raman spectra of human keratotic biopolymers: skin, callus, hair and nail”, J. Raman Spectrosc. 25, 95-98 (1994).
- A. C. Williams, B. W. Barry, H. G. M. Edwards and D. W. Farwell, “A critical comparison of some Raman spectroscopic techniques for studies of human stratum corneum”, Pharmaceutical Research 10, 1642-1647 (1993).
- P.J. Caspers, G. W. Lucassen, R. Wolthuis, H. A. Bruining and G. J. Puppels, ” In vitro and in vivo Raman spectroscopy of Human Skin”, Biospectroscopy 4, S31-S39 (1998).
- R. R. Anderson, J. Hu and J. A. Parrish, “Optical radiation transfer in the human skin and applications in in vivo remittance spectroscopy”, in Bioengineering and the Skin, R. Marks and P. A. Payne, Eds. (MTP Press, Lancaster, England, 1981).
- P.L. Walling and J. M. Dabney, “Moisture in skin by near-infrared reflectance spectroscopy”, JSCC 40, 151-171 (1989).
- K. Martin “In vivo measurements of water in skin by near-infrared reflectance”, Appl. Spec. 52, 1001 (1998).
- M. Joy and D. M. Lewis, “The use of Fourier transform infrared spectroscopy in the study of the surface chemistry of hair fibres”, Int. J. Cosmet. Sci. 13, 249-261 (1991).
- V. Signori and D. M. Lewis, “FTIR investigation of the damage produced on human hair by weathering and bleaching processes: Implementation of different sampling techniques and data processing”, Int. J. Cosmet. Sci. 19, 1-13, (1997).
- J-L. Bantignies, G. Fuchs, G. L. Carr, G. P. Williams, D. Lutz and S. Marull, “Organic reagent interaction with hair spatially characterized by infrared microspectroscopy using synchrotron radiation”, presented at the 20th IFSCC Congress, 1998.
- L. E. Jurdana, K. P. Ghiggino and K. W. Nugent, “Confocal laser Raman microprobe studies of keratin fibres”, Text. Res. J. 65, 593-600 (1995).
- C. M. Pande, “FT-Raman spectroscopy-Applications in hair research”, JSCC 45, 257-268 (1994).
- W. Akhtar, H. G. M. Edwards, D. W. Farwell and M. Nutbrown, “Fourier-transform Raman spectroscopic study of human hair”, Spectrochimica Acta Part A 53, 1021-1031 (1997).
- Y. Ozaki, T. Miura, K. Sakurai and T. Matsunaga, “Nondestructive analysis of water structure and content in animal tissues by FT-NIR spectroscopy with light-fibre optics. Part I: Human hair”, Appl. Spec. 46, 875-878 (1992).
- H. M. Klimisch, G S. Kohl and J. M. Sabourin, “A quantitative diffuse reflectance method using Fourier transform infrared spectroscopy for determining siloxane deposition on keratin surfaces”, JSCC 38, 247-262 (1987).
- E. Dreassi, G. Ceramelli, P. Mura, P. L. Peruccio, F. Vocioni, P. Bartalini and P. Conti, “Near-infrared reflectance spectrometry in the study of atopy. Part 2. Interaction between the skin and polyethylene glycol 400, isopropyl myristate and hydrogel”, Analyst 122, 771-776 (1997).
- K. A. Martin, “Direct measurement of moisture in skin by NIR spectroscopy”, JSCC 44, 249-261 (1993).
- M. Gloor, U. Willebrandt, G, Thomer and W. Kupferschmid, “Water content of the horny layer and skin surface lipids”, Arch. Dermatol. Res. 268, 221-223 (1980).
- M. Gloor, B. Heymann and Th. Stuhlert, “Infrared spectroscopic determination of the water content of the horny layer in healthy subjects and in patients suffering from atopic dermatitis”, Arch. Dermatol. Res. 271, 429-436 (1981).
- R. O. Potts, “In vivo measurement of water content of the stratum corneum using infrared spectroscopy: A review”, Cosmet. and Toil. 100, 27-31 (1985).
- R. O. Potts, D. B. Guzek, R. R. Harris and J. E. McKie, “A non-invasive, in vivo technique to quantitatively measure water concentration of the stratum corneum using attenuated total-reflectance infrared spectroscopy”, Arch. Dermatol. Res. 277, 489-495 (1985).
- P. A. D. Edwardson, M. Walker, R. S. Gardner and E. Jacques, “The use of FT-IR for the determination of stratum corneum hydration in vitro and in vivo”, J. Parmaceut. and Biomed. Anal. 9, 1089-1094 (1991).
- J. deRigal, M.J. Losch, R. Bazin, C. Camus, C. Sturelle, V. Descamps and J. L. Leveque, “Near-infrared spectroscopy: A new approach to the characterization of dry skin”, JSCC 44, 197-209 (1993).
- E. Dreassi, G. Ceramelli, L. Fabbri, F. Vocioni, P. Bartalini and P. Conti, “Application of near-infrared reflectance spectrometry in the study of atopy. Part 1. Investigation of skin spectra”, Analyst 122, 767-770 (1997).
- M. G. Sowa, J. Wang, C. P. Schultz, M. K. Ahmed and H. H. Mantsch, “Infrared spectroscopic investigation of in vivo and ex vivo human nails”, Vibrational Spectrosc. 10, 49-56 (1995).
- C. R. Robbins, Chemical and Physical Behavior of Human Hair, 3rd Ed., Springer-Verlag, New York, 1994.
- L. D. Rhein, F. A. Simion, C. Froebe, J. Mattai and R. H. Cagan, “Development of a stratum corneum lipid model to study the cutaneous moisture barrier properties”, Colloids and Surfaces 48, 1-11 (1990).
- G. M. Golden, D. B. Guzek, A. H. Kennedy, J. E. McKie and R. O. Potts, “Stratum corneum lipid phase transitions and water barrier properties”, Biochemistry 26, 2382-2388 (1987).
- D. Bommannan, R. O. Potts and R. Guy, “Examination of stratum corneum barrier function in vivo by infrared spectroscopy”, J. Invest. Dermatol. 95, 403-408 (1990).
- M. Denda, J. Koyama, R. Namba and I. Horii, “Stratum corneum lipid morphology and transepidermal water loss in normal skin and surfactant-induced scaly skin”, Arch. Dermato. Res. 286, 41-46 (1994).
- E. G. Bendit, “Infrared absorption spectrum of keratin. I. Spectra of a -, b – and supercontracted keratin”, Biopolymers 4, 539-559 (1966).
- C. B. Baddiel, “Structure and reactions of human hair keratin: An analysis by infrared spectroscopy”, J. Mol. Biol. 38, 181-199 (1968).
- L. E. Jurdana, K. P. Ghiggino, I. H. Leaver, C. G. Barraclough and P. Cole-Clarke, “Depth profile analysis of keratin fibres bt FT-IR photoacoustic spectroscopy”, Appl. Spec. 48, 44-49 (1994).
- E. Hoting, M. Zimmermann and S. Hilterhaus-Bong, “Photochemical alterations in human hair. I. Artificial irradiation and investigations of hair proteins”, JSCC 46, 85-99 (1995).
- S. Ratnapandian, S. B. Warner and Y. K. Kamath, “Photodegradation of human hair”, J. Cosmet. Sci. 49, 309-320 (1998).
- S. Hilterhaus-Bong and H. Zahn, “Contributions to the chemistry of human hair. I. Analyses of cystine, cysteine and cystine oxides in untreated human hair”, Int. J. Cosmet. Sci. 9, 101-109 (1987).
- J. Strassburger and M. Breuer, “Quantitative Fourier transform infrared spectroscopy of oxidized hair”, JSCC 36, 61-74 (1985).
Received in original format 21st April 1999, accepted 4th May 1999.