The use of Diamond as an ATR material
3. The use of Diamond as an ATR material
Dave Coombs
Graseby Specac,
River House, 97 Cray Avenue,
St Mary Cray, Orpington,
Kent, BR5 4HE
United Kingdom
Telephone: +44 (0) 1689 873134
Fax: +44 (0) 1689 878527
E-mail: coombsd [at] compuserve.com
Introduction
The development of Diamond as an ATR material has opened up a number of sampling opportunities in the mid infrared. Diamond has a number of properties important for the infrared spectroscopist. The high refractive index ensures that for incidence angles of 45 degrees, a typical infrared penetration of around 2 microns is achieved. Consequently, single reflection measurements are capable of yielding good quality spectra under conditions of good optical contact with the ATR crystal.
The extraordinarily stable covalent bonding structure of diamond accounts for its hardness and physical strength .The principal benefit is that ATR sampling for abrasive or chemically hostile samples is now routine. The large band gap between the valence and conduction electrons in diamond means that radiation of a very broad wavelength range is able to pass through almost unhindered. Diamond therefore is able to transmit from the UV-Visible to the far infrared. There are mid infrared lattice vibrations at around 2000 cm-1 however careful optical design and short path lengths through the diamond ensure that ATR accessories perform well in this region of the spectrum and that the full transmission capabilities of diamond are properly utilised.
Finally, the high frequency vibrations of the carbon atoms in the lattice impart yet another useful property open to exploitation by the optical engineer. Heated ATR experiments are straightforward because of the high thermal conductivity of diamond. In fact diamond is five times more efficient at conducting heat than even copper which ensures the place of this unique material in the infrared hall of fame.
A reminder of the nature of Attenuated Total Reflectance spectra
In a simple transmission experiment, the light beam enters the sample normal to its surface and passes through. If we change the incidence angle to one other than zero degrees, the sample would reflect much of the incident radiation depending upon its actual reflectivity. This forms the basis for specular reflectance measurements. If an IR transparent crystal such as diamond is placed behind and in good optical contact with the sample, light passing into the diamond can be made to emerge on the opposite side having made a reflection at the diamond-sample interface. In fact the emerging light contains important absorbance information about the sample. There are certain geometric requirements for this and importantly the refractive index of the crystal must exceed that of the sample.
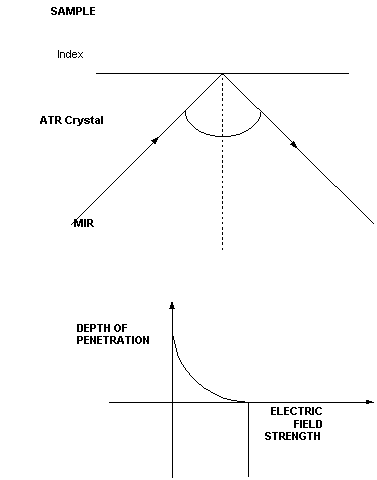
Figure 1. The ATR Experiment
Regarding the optical and geometrical requirements for this process, we need to consider the differences in the refractive indices between the two materials and the angle of incidence of the radiation. At the first contact between the radiation and the sample, a fraction will be reflected and a fraction transmitted. When light passes through two media in intimate optical contact with each other and having different refractive indices, the path of the light will be distorted depending on the incidence angle. The incident light must therefore be below some critical angle in order to set up the process. In fact the situation is a little more complex than this because the light is polarised perpendicular and parallel to its direction of travel and the reflectivity is different for each of these polarisation’s and also varies with incidence angle. # ATR is a surface sensitive technique. Considering the refractive index of diamond and a typical organic polymer to be 2.4 and 1.5 respectively, the top two microns or so of the surface will be sampled at 45º incidence. Finally, this sampling depth changes in two fundamental ways. It decreases as the refractive index of the crystal increases and it also decreases as the incidence angle is increased. Also explained in IJVS Vol I, 438-444 (1997) – Editor.
# Editor’s Note: This property is discussed further in the next article.
Some experimental aspects of choosing diamond as a good ATR crystal.
A key experimental requirement is that a smooth flat scratch free sampling surface is maintained in order to maximise both contact and signal to noise ratio performance for the analysis. A uniform pressure distribution is also required. This is more easily achieved using a robust, well supported small surface area diamond. Reproducibility is also easier than with solids sampling using the bulkier classical 6 reflection ZnSe type ATR units. These larger ZnSe units also have a more open architecture with a wide ray acceptance angle cone. Diamond has a smaller acceptance angle cone and requires good optical design in order to avoid working close to the critical angle whilst enjoying a compact beam condensing arrangement in the accessory. The high load bearing capacity of diamond allows for (near) perfect optical contact between the sample and diamond and results in good spectra of even the most intractable of sample types.
Design characteristics for a diamond ATR unit
Many of the ideal design characteristics for a diamond ATR unit are incorporated into the ‘Golden Gate’ single reflection diamond ATR accessory manufactured by Specac, Orpington, Kent, UK. A type IIa industrial grade single diamond crystal is high temperature metal bonded into a Tungsten carbide support disc at 1000℃ (Figure 2). This ensures the most durable robust and permanent mounting arrangement possible and has no dependence on adhesive bonding which could otherwise result in the removal of the diamond when cleaning with certain solvents.

Figure 2. Diamond in its mount
Samples are forced into almost perfect optical contact with the diamond using a sapphire anvil assembly mounted overhead on a unique swing bridge. Reproducible loads of 3 kbar can be applied using a special torque wrench supplied with the unit. The anvil is fully interchangeable with a series of alternative special anvils. These include a stainless steel anvil for polymer pellets, grooved anvils for analysing polymer coatings on wires, and an anvil for studying air sensitive or reactive compounds.

Figure 3. Golden Gate ATR Diamond accessory
In this application, the top plate of the accessory may be removed to a glove box or controlled atmosphere and the sample loaded into position and sealed using a special anvil in order to protect it from the external environment. The top plate is subsequently replaced ready for the measurement. A micro reaction/flow cell anvil is also fitted in a similar manner and can be used to study low volume (28 microlitres) liquid flows or static injections up to 1000 psi.
The diamond ATR top plate is mounted onto a fully enclosed beam condensing optics box used to condense the IR beam by a factor of 4X. Typically this results in a sensitive sampling area of approximately 1mm in the centre of the diamond crystal. The optics are robust and maintain their stability of alignment and can also be purged with dry air or nitrogen. The beam condensation is achieved using the more optically efficient lens option in combination with mirrors. The lenses are made of ZnSe, which transmits, to around 650cm-1. Alternatively KRS-5 lenses can be used for transmitting to longer wavelengths (300 cm-1). The small sample hot spot makes the accessory ideal for microsampling.
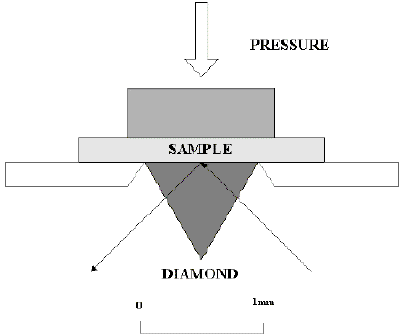
Figure 4. Schematic Diamond ATR
Single reflection diamond ATR has been successfully applied in a number of micro-sampling applications. Particularly, single polymer fibres as small as 20 microns have been analysed with the technique and other forensic type applications such as paint fragments and particulates can yield good quality spectra without the need for cumbersome sample preparation. A benefit of this approach is that qualitative analyses can be quickly performed which can be used as a screening technique before engaging in the more expensive and time consuming use of infrared microscopy. A combination of the two makes a real contribution towards cost effective and efficient analysis of small samples.
Single pellets, powders, coated wires, intractable or opaque solids, corrosive liquids, curing polymers, and air sensitive compounds can now be analysed with one multi purpose accessory.
Many reactions and processes occur at high temperatures. The heated version Diamond ATR top plate allows for all the benefits of the standard unit up to 200℃.
The uniquely high thermal conductivity of diamond in combination with the low thermal mass top plate, ensure that the high power heaters built in to the plate in close proximity to the diamond, leads to both rapid and efficient heating of the diamond. The heated unit is used with an electronic temperature controller with digital readout to 1℃. Some applications for this unit have been polymerisation reactions, cooking processes, degradation/decomposition, and phase transitions. A special reaction cell version of the diamond ATR can be configured for the study of low volume (24ml) reaction studies and can incorporate flow, mixing, options at combinations up to 200℃ at 3000psi. This unit has proved ideal for small scale optimization of process parameters, for the studies of highly acidic and caustic solutions and for the study of slurries with abrasive particulates in suspension.
4. Some applications examples for
single reflection Diamond ATR
We have included some typical applications for this technology to illustrate the versatility of using just a single reflection from a diamond ATR crystal. Remember these spectra all correspond to a sample penetration of just two microns. This shows the awesome capability of using Diamond, as you may never have thought possible before!
Acid catalysed esterification of methanol in the presence of sulphuric acid
Liquids have never posed a problem for the ATR technique except when they contain acids that would attack conventional ATR crystals such as ZnSe. The data shown in Figure 5 was recorded at room temperature using a sample volume of less than 30 microlitres.
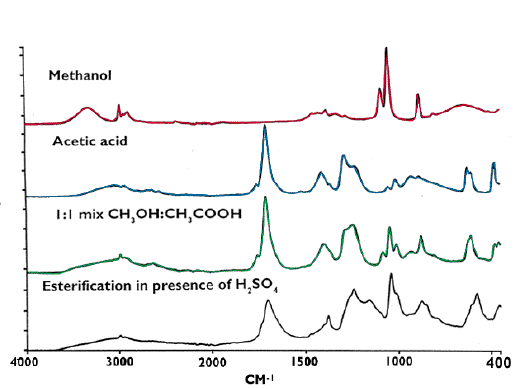
Figure 5. ATR Infrared spectra of a reacting system.
The bottom spectrum is the product.
Understanding the process of cement curing
You may well have seen spectra of paint drying in the past but here is a novel variation on a theme. Cement is mineralogically complex and so too are the reactions involved in the cure process. Even more demanding is understanding how cement minerals decompose under conditions of high temperature and pressure. This is especially important in the oil industry. Spectra shown in Figure 6 were recorded at high temperature and illustrate the decomposition of the mineral Ettringite in the presence of water under sealed conditions. Classically, X-ray diffraction techniques would have been used in crystallographic studies of these systems. Infrared however has an important complementary role in yielding information on how water is involved – a study not possible before.
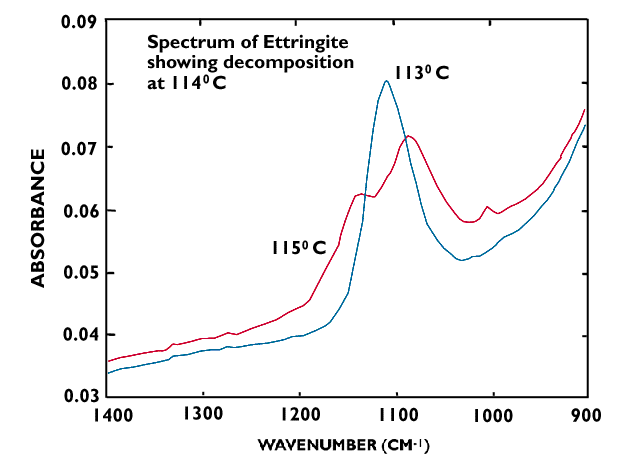
Figure 6. The FTIR-ATR spectrum of ettringite
showing decomposition at 114°C
What happens when bread stales?
There are a number of schools of thought on this one! Spectra of fresh bread show distinct features attributed to water, fat, starch and protein (Figure 7). When the bread stales there is good evidence for a reduction in the water content and it would appear that a subtle difference in the starch region of the spectrum may account for a change in the crystal structure of the starch with its corresponding effect on texture!(Figure 8)
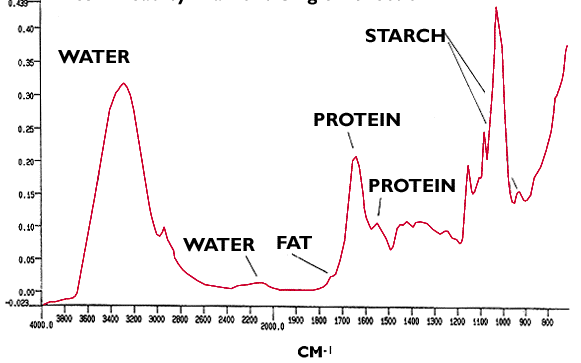
Figure 7. Fresh Bread by Diamond single reflection ATR
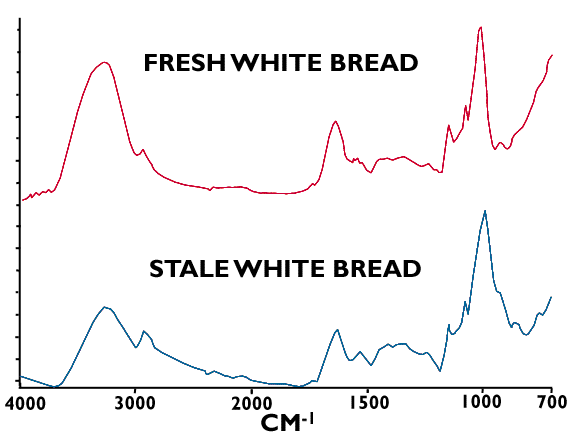
Figure 8. ATR Infrared spectra of fresh and stale bread
Surely not…. it cannot be that easy to obtain an infrared spectrum of oleum?
Sulphuric acid is a strong dibasic acid with ample capability of digesting standard ATR materials. The oleums are sulphuric acid/sulphur trioxide cocktails with enormous industrial importance. This includes the manufacture of important nitrogenous chemicals such as nitro-cellulose and various dyestuffs. They are also used in the purification processes for petroleum products. The spectrum shown was recorded using the most minute pool of liquid imaginable…and why not!(Figure 9).
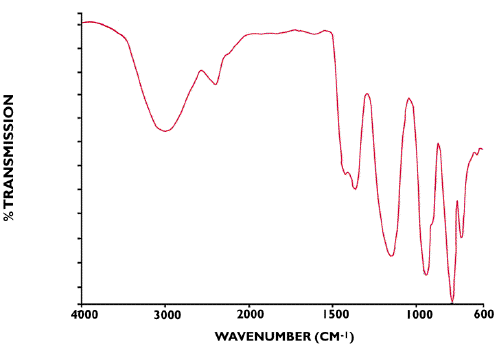
Figure 9. ATR Infrared spectra of oleum H2SO4+SO3
Are you sure that it was my car that you claim ran into you?
These spectra shown in Figure 10 were recorded in a real forensic crime laboratory. They illustrate the data obtained from a real vehicle interaction in a case where the suspect failed to stop. These fragments were approximately 0.5 mm in diameter and the data was recorded using a DTGS detector. Undoubtedly a big success for three minutes analysis time.
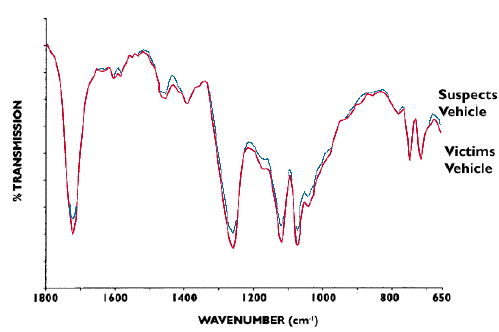
Figure 10.
We will be pleased to show also, other examples of data we have recorded using single reflection diamond ATR .If you have a particular interest not mentioned in this short applications brief, then please contact me via one of the routes below and I will be pleased to help.
Editor’s Note:
So, there you have it. Until January I had never used a diamond ATR accessory. I then became involved in a project to examine a very wide range of polymers and I was staggered at the convenience. I found that Davids’ claim above is real - almost anything, a powder, lump or film placed on the horizontal diamond surface and pressurised, gave an excellent spectra. The ease and speed of sampling is truly impressive. Of course, most of the comments David makes apply equally to systems from other competing manufacturers.
