Spotlight – Perkin Elmer’s NEW Mid Infrared Microscope Imaging System
Robert Hoult and Richard Spragg
Perkin Elmer Ltd
Seer Green
Buckinghamshire
HP9 2FX, UK
There are two conventional ways of generating mid infrared frequency specific images on a microscope.
- To record spectra point by point across the area of interest and then assemble the image at any frequency you select. Using filters or diffraction grating monochromators it is possible to record transmission or reflection data at fixed frequencies again point by point. Both methods work, the latter is more versatile in that many images at difference frequencies can be recovered from the recorded data once it is placed on memory, but it is very, very slow. The alternative – the use of filters is much faster but the data must be recorded again and again at several frequencies as required.
- The image from the microscope is focussed onto an array detector. The microscope is illuminated with the interferogram from an FTIR hence each element of the array records a different interferogram. In this case, the image is available at a wide range of frequencies sampled simultaneously.
The Array detector method is superb but VERY expensive. Most of the appropriate detector elements have been developed for military hardware where cost is no object. Need we say more?
The Perkin Elmer team has addressed this problem using a fairly conventional infrared microscope, a commercial Spectrum One FTIR and a LINEAR array detector resulting in a high performance system at a sensible price.
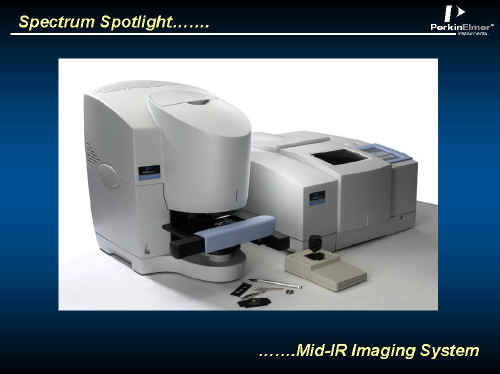
Figure 1.
The System
Light from the Spectrum One interferometer passes into a redesigned version of Perkin Elmers’ Autoimage Microscope.
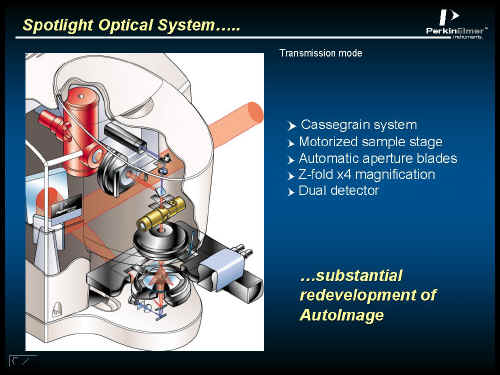
Figure 2.
The radiation path is shown in orange. The optics are of the Cassigrainian Mirror type (no lenses are used). Once imaged onto the sample held on the stepper motor controlled stage, the collected radiation passes through a Z-fold x 4 magnifier (the horizontal gold coloured unit in the centre of Figure 2) – more to follow – and then to the detector – the red/brown unit at the top. This contains 2 elements – a conventional single point detector and a linear array. A conventional visible light path is also supplied so that the user can ‘see what he or she is doing’.
The detector is very unusual and is illustrated below.
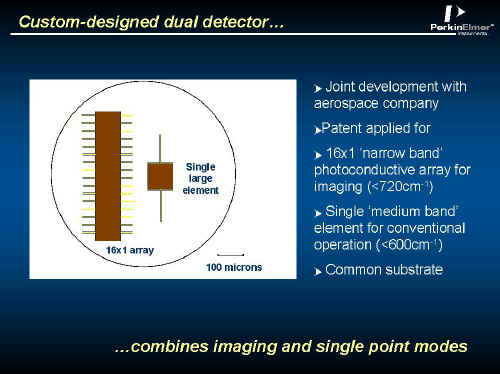
Figure 3.
Within an area of about 1 sq. mm, the two detectors are arranged side by side each other, each pixel being hardwired to the outside of the evacuated and cooled detector enclosure.
The microscope can operate in two ways – point by point or strip by strip, the switch from one to another being carried out using motorised aperture blades in the optical path. You can see the blades in Figure 2 just above the Z fold magnifier.
The effect on the detector elements is illustrated below in Figure 4, whilst the way the image is scanned as shown in Figure 5. The 16 interferograms are recorded, the stage is moved, the next set is recorded and so on.
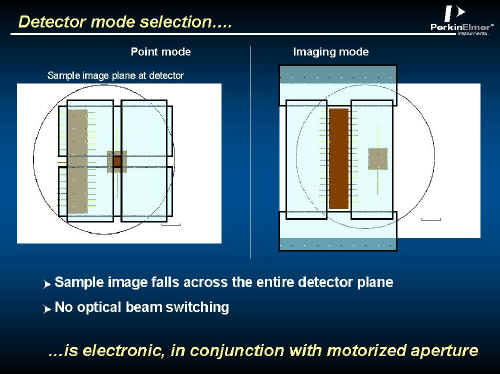
Figure 4.
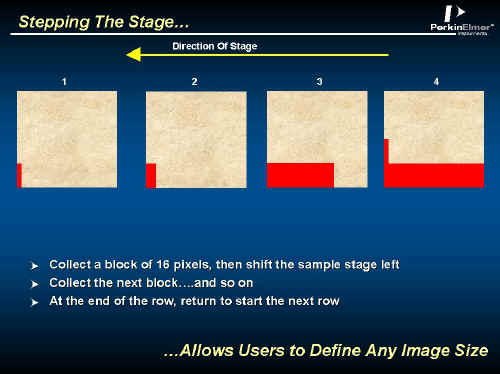
Figure 5.
To regularise and maximise performance it is obvious that the movement of the stage should occur only at the ends of the interferometer scan where no useful data is being recorded. See Figure 6. Note that by scanning the interferometer rapidly, enormous amounts of data can be accumulated very quickly.
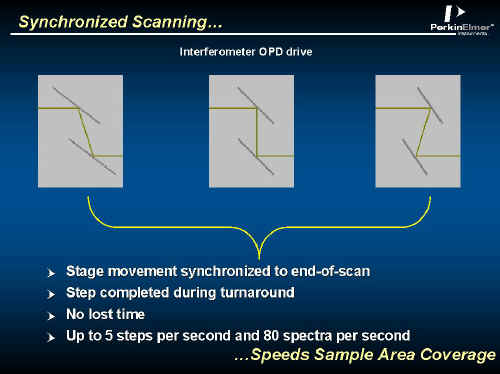
Figure 6.
Z Fold 4 x Magnification
To improve versatility in the system, a small path extending system is incorporated into the optical path. In one orientation, light goes straight through but rotate the tubular assembly about its axis and the optical path is diverted through a combination of mirrors increasing the microscope’s magnification by four times. The effect is that each pixel views a patch ~6.25µ square at the sample instead of the normal ~25 µ square. So, the user can have high spatial resolution or look at lower resolution over a wider area. See Figure 7.
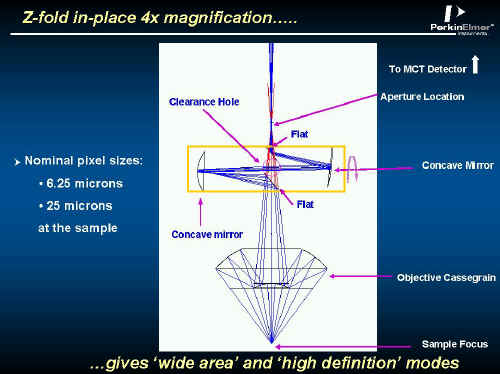
Figure 7.
Examples of application of Spotlight
In Figure 8, you will see in grey, a view of a tiny polypropylene specimen containing an inclusion. The identification of bits and pieces of rubbish in otherwise uncontaminated material is frequently encountered in polymer processing. The identity can lead to the origin of the inclusion and is commercially crucial. On the lower right part of Figure 8, you will see a pseudo 3D image of the defect recorded in only 2 minutes. The sequence of operations required to generate the results is illustrated in Figure 9. Note – the ‘background’ must be appropriately recorded (as you always do in FTIR). The coloured picture to the right develops in real time. The x and y axes are distance, the colour goes from blue?red as the absorbance increases.

Figure 8.

Figure 9.
In Figures 10, 11, 12 and 13 you will see data from a fingerprint on a glass slide. These spectra were recorded in transmission using the region above 2000 cm-1. The striking thing about this image is its size, over 400,000 spectra! Measuring this took about one and a half hours. The average spectrum in Figure 11 shows weak CH2 and OH absorptions. In Figure 12 an image at a CH2 frequency follows the ridges of the fingerprint, but an image of the OH highlights isolated spots. Closer examination in Figure 13 reveals that the spectrum at one of the spots has CH3, CH2, NH and OH absorptions typical of proteins, while neighbouring spectra just show finger grease. Forensic applications of this type of analyses are obvious!

Figure 10.
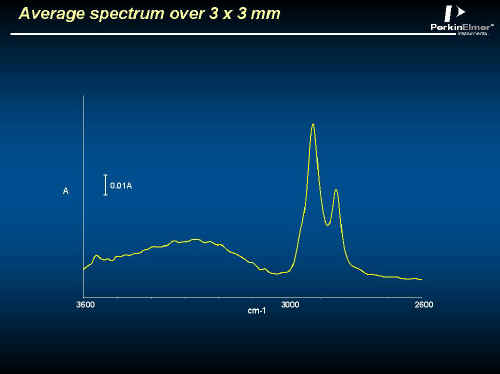
Figure 11.
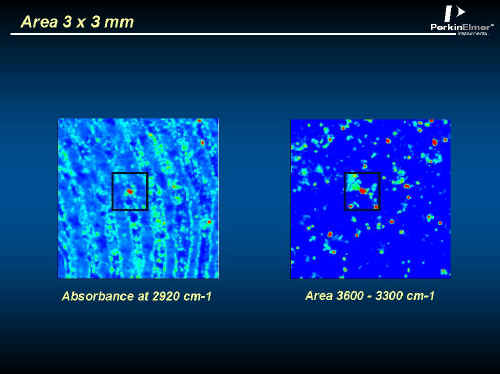
Figure 12.
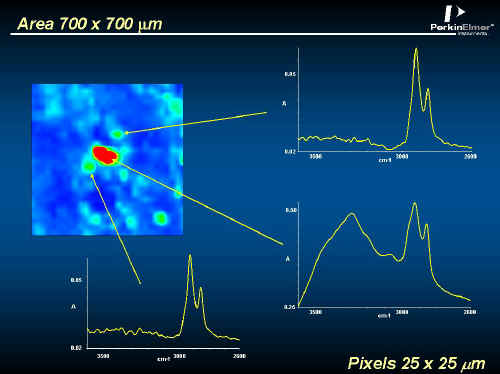
Figure 13.
Orientation of the polymer in a PET bottle
The bottles we all buy containing carbonated drinks like Coke are made of polyethylene terephthalate PET.
Now PET is very strong, particularly if the molecules are aligned. Like most regular polymers, PET can be extended in one direction to align most of the chains parallel to one another. Unusually, it is quite simple to extend the polymer in two orthogonal directions – the chains align and the benzene rings rotate until they are parallel to one another and are packed on top of each other. Highly oriented PET is strong, very resistant to tearing, hard and very impermeable to CO2 – ideal for a Coke bottle. The bottles are made by moulding a small bottle (around 20mls capacity and called a ‘pre-form’), heating it to more than 100ºC and then blowing with heated air. The bottle expands into a heated mould and hey presto you have your bottle. A complex process like this is fraught with potential disasters. Uneven heating or blemishes in the pre-form will produce an unacceptable product with inadequate molecular orientation. One of the best and most convenient ways to monitor orientation is to use FTIR with a polarizer [see ijvs – Volume 5, Edition 1] Below, we give an example of an orientation measurement using a polarizer in the Spotlight system. Figure 14 shows the conventional spectrum of the wall of a PET bottle. The problem here is that the wall is far too thick for absorption experiments. PET shows ‘dichroism’ – different results when the electric vector of the IR is || or to the draw (or molecular) axis. See Figure 15 where spectra are shown in a thin film only 40µ thick.
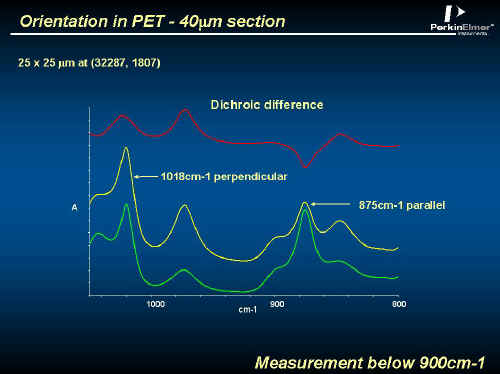
Figure 14.
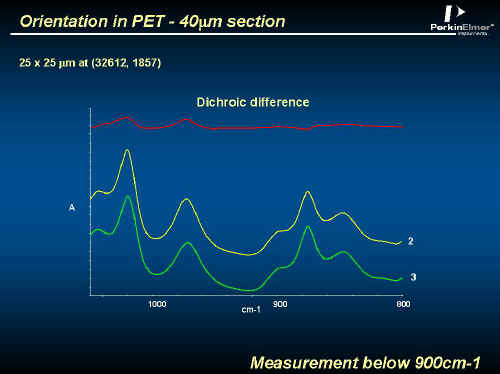
Figure 15.
The bottle wall shows a perfectly reasonable spectrum in the NEAR IR (Remember, the extinction coefficients typical in the NIR are much lower than in the mid IR). See Figure 16. The band areas can be related to orientation. See Figure 17.
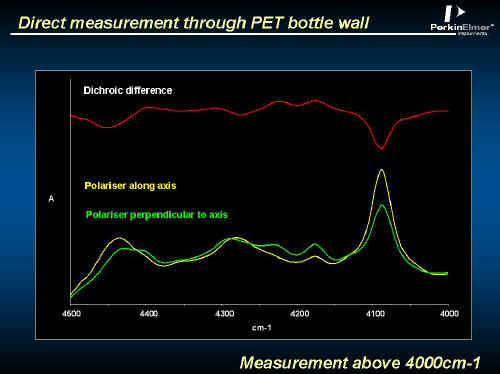
Figure 16.
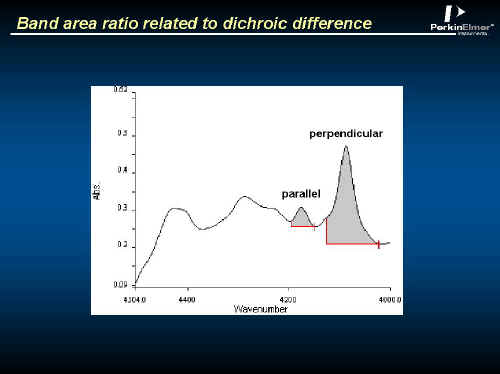
Figure 17.
Quite obviously, the orientation of a bottle will vary within the bottle, Figure 18 shows the spatial distribution based on Figure 16 and 17. Again this is an enormous image containing over 600,000 spectra that took just over two hours to record..
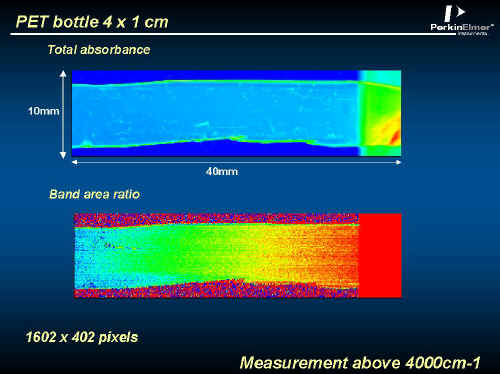
Figure 18.
Sectioning the wall of the bottle to produce thin samples is not very practical, but specular reflection off the surface of the PET is an alternative. The spectral reflectance spectrum can be recorded in polarized light and the spectrum is highly specific just like its transmission analogue. The snag is that the spectral reflectance spectra are weak.
In Figure 19, the polarized specular reflectance spectrum of a PET bottle is shown. The inside surface is not the same as the outside – see Figures 19 and 20. Using the Kramers Kronig algorithm, the absorbency component can be extracted so that this spectrum looks like conventional absorption spectra – See Figures 21 and 22. Clearly, the inner surface is better oriented than the outer.
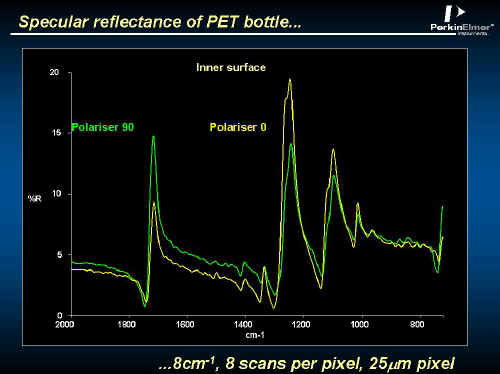
Figure 19.
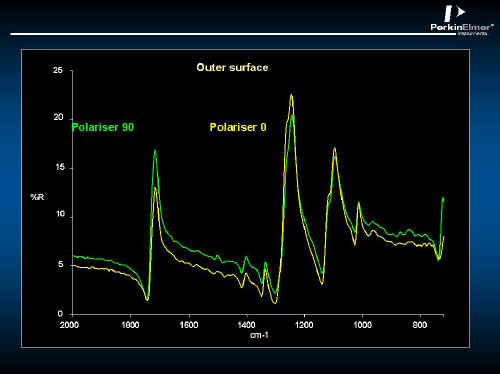
Figure 20.
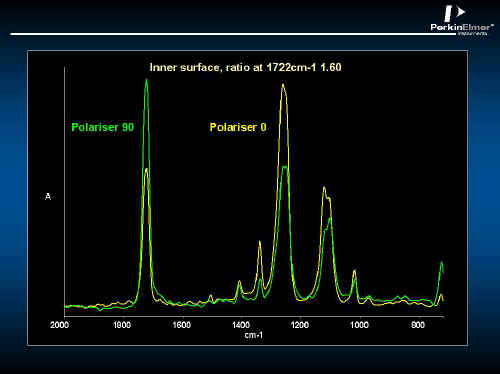
Figure 21.
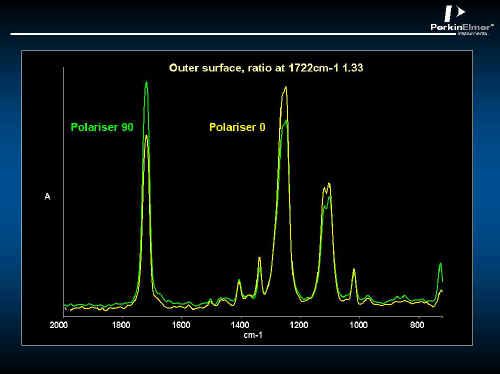
Figure 22.
REF: R. Hoult & R Spraggi, Int. J. Vib. Spect., [www.irdg.org/ijvs] 6, 2, 4 (2002)