IR cell reactors for in situ studies
J. Ryczkowski
University of Maria Curie-Sklodowska,
Faculty of Chemistry
Department of Chemical Technology,
Pl. M. Curie-Sklodowskiej 3,
20-031 Lublin, Poland
tel. +48-81 537-55-96
fax: +48-81 537-55-65
E-mail://ryczkows [at] hermes.umcs.lublin.pl">ryczkows [at] hermes.umcs.lublin.pl
Introduction
Catalysis play a key role in nature and society since almost every reaction requires a catalytic material. Catalysts facilitate a chemical reaction by lowering the energy barrier of the reaction pathway and thus increasing the reaction rate. Monitoring the events taking place in such materials is crucial for understanding the reaction mechanisms of many important chemical processes and would allow the rational design of new or better catalytic solids. This monitoring includes the observation of reaction intermediates, the discrimination between spectator species and active sites, the quantification of unusual oxidation states and coordination environments of metal ions in catalyst materials as well as the migration and mobility of species at the catalyst surface. This is the field of in situ spectroscopy where in situ refers to the study of catalytic materials at their working place under real reaction conditions; e.g, in a gas stream of reactants and at high temperatures. Researchers are nowadays working to develop analytical tools that allow them to follow the physicochemical processes taking place in an active catalyst in real time and under operating conditions; i.e., they are using in situ characterization techniques to understand the working of catalyst materials. Catalysis is primarily an applied science, however, and as such should reasonably be expected to provide major assistance in reaching the goals of better catalysts and improved catalytic processes all arising from a better fundamental understanding of catalyst surface chemistry. This is an area in which IR will undoubtedly make further major contributions.
After nearly 60 years of intensive application, infrared spectroscopy (IR) remains the most widely used, and usually most effective, spectroscopic method for characterization of surface chemistry of heterogeneous catalysts [1,2]. From the historical point of view both catalysis and IR spectroscopy are of the same “age”. Scientific bases for those both scientific areas were created at the beginning of XIX century, and since 1940 they are successfully “co-operating” together [1]. In the past few years one can observe a growing interest in the application of IR techniques in catalytic investigations. One of the reasons, among the others, is their wide distribution (nowadays, IR and/or FT-IR spectrometers belong to the standard equipment of every scientific laboratory) and the relatively low costs (compared to the other modern physico-chemical techniques for surface characterization) of the base instrument.
A variety of IR techniques has been and can be used in order to obtain information on the surface chemistry of different solids (Figure 1). Special meaning have investigations carried out under the reaction conditions. In principle for in situmeasurements, all forms of IR spectroscopy are suitable. For most practical experimental reasons, however, the transmission-absorption and diffuse reflectance techniques are best suited. This is more related to the design of the cells that are to be used as reactor, rather than with the principal problems of the other techniques.
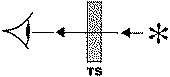
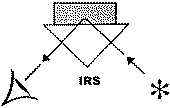
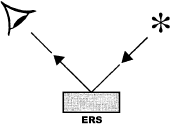
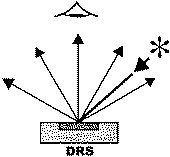
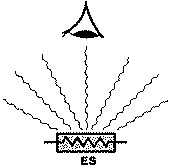
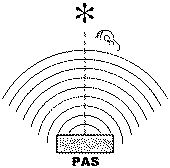
Figure 1. Basic spectroscopic techniques: TS – transmission spectroscopy, IRS – internal reflection spectroscopy (or attenuated total reflectance – ATR), ERS – external reflection spectroscopy, DRS – diffuse reflectance spectroscopy, ES – emission spectroscopy, PAS – photoacoustic spectroscopy [3]
Cells for in situ studies
The IR cell in which the catalyst sample is pre-treated and subsequently studied is extremely important in surface studies. The perfect, all-purpose cell has yet to be devised, and cell design is normally chosen to suit the purposes of a particular study. Some features are usually of overriding importance in a given application. If catalytic reactions are to be studied the exposure of catalytic metals must be eliminated in cell construction, and bare heating elements within the cell are ruled out. In some surface characterisation studies such features may be completely acceptable, but even in such studies it is well to avoid any possibility of Ni or other carbonyls being formed from cell components. A variety of relatively simple, but effective, cells has been used for studies. Many of these have been described in the literature and schemes of them have been given [4].
As it was mentioned, starting from the pioneering work of Eischens et al. [5] on supported metal catalysts (adsorption of ammonia and carbon monoxide), the use of IR in surface science and catalysis has grown rapidly. IR, with its high-energy resolution, can be a very appropriate tool to investigate the internal and external modes of adsorbates and their vibration dynamics. The development of in situ vibrational spectroscopies applicable to metal-support interfaces in recent years has exerted a profound influence on our understanding of adsorptive chemistry in heterogeneous systems. Some pertinent information can be obtained from the number of bands in the spectra at a single stage of surface coverage as shown in the original spectra of chemisorbed CO. However, these single stage spectra do not reveal the relative strength of bonding for the chemisorbed CO contributing to each band or the effect of interaction on the band positions. To obtain this information the spectra of chemisorbed CO were studied as a function of surface coverage over silica-supported Pt, Pd and Ni [6]. The authors wrote: “In order to carry out this work efficiently it was necessary to design apparatus in which the IR spectra could be obtained while the samples were subjected to a wide range of temperatures and pressures. Successful development of this in situ apparatus not only makes it possible to study the effect of surface coverage but also opens the way to IR studies of chemisorbed molecules while reactions are in progress” [6]. It was one of the first in situ cells for IR studies published in scientific literature.
In catalytic research very often “home-made” IR cell reactors are used for the particular in situ studies. In the following figures there are presented selected examples of the “home-made” and commercially produced such devices. Many of them are successfully used in the research for a considerable period of time. For the figures presented only general information will be provided. Details can be found in the quoted references.
Peri and Hannan [7] designed an IR cell for the determination of the surface hydroxyl groups on g-alumina (Figure 2).
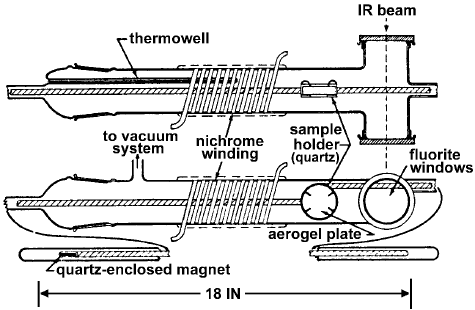
Figure 2. Infrared cell [7].
Greenler [8] has adopt his cell for IR study of the adsorption of methanol and ethanol on aluminum oxide (Figure 3).
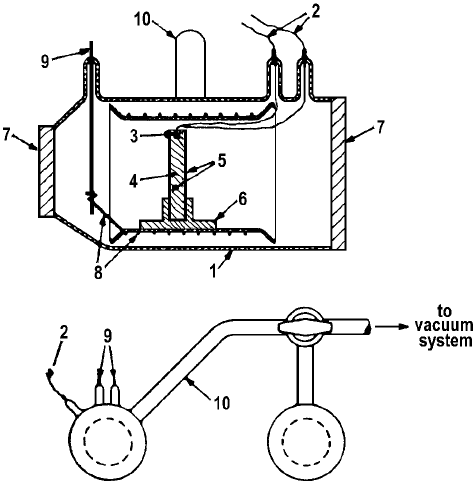
Figure 3. Diagram of the sample cell, showing: 1 – Pyrex cell body, 2 – thermocouple leads, 3 – thermocouple hole in sample support disk, 4 – sample support disk, 5 – alumina sample, 6 – sample holder, 7 – cell windows, 8 – platinum heater wire, 9 – tungsten wire for heater leads, 10 – connecting tube to vacuum system [8].
Ryason [9] has described a stainless steel quartz cell for IR transmission spectroscopy of catalyst wafers (Figure 4).
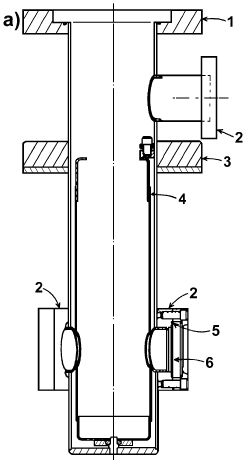
Figure 4. IR cell for in situ studies: A) cross section of infrared cell: 1 – Conflat flange, 2 – Conflat flange, 3 – rotable magnet, 4 – rotable heat shield, 5 – Viton gasket, 6 – alkali halide window; . B) cell insert: a, b – Conflat flanges, c – stainless steel-Pyrex Housekeeper seal, d – fused silica tube, e – silica thermocouple tube bundle, f – thermocouple junction holder, g – holes for thermocouple wires, h – thermocouple shields, i – platinum wire furnace, j – removable silica wafer holder [9].
Amenomiya [10] has described a double beam cell for the high temperature infrared spectroscopy of adsorbed species during catalytic reaction (Figure 5).
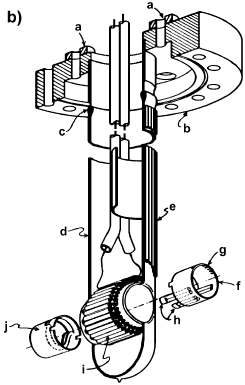
Figure 5. IR cell: 1 – catalyst, 2 – furnace block, 3 – heater element, 4 – thermocouple, 5 – Cajon fitting [10].
Hicks et al. [11] have reported on the design and construction of a reactor for in situ IR studies of catalytic reactions (Figures 6 and 7). It seems, that this is a very successful design because it is extensively used by Bell and co-workers for more than 20 years.
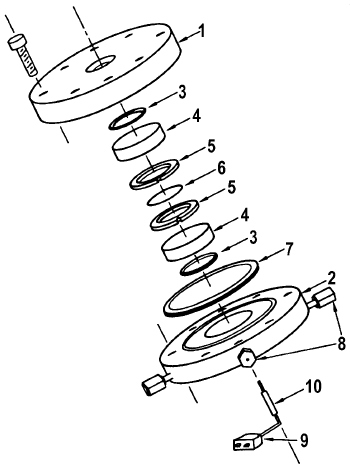
Figure 6. Exploded view of the IR reactor: 1 – top flange, 2 – bottom flange, 3 – Kalrez O-ring, 4 – CaF2 window, 5 – sample holder, 6 – catalyst disk, 7 – copper gasket, 8 – Swagelok fitting, 9 – sheathed thermocouple, 10 – sleeve attached to thermocouple sheath [11].
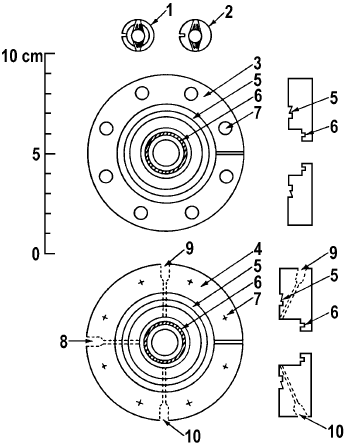
Figure 7. Details of IR reactor and sample holder: 1 – bottom half of sample holder, 2 – top half of sample holder, 3 – top flange, 4 – bottom flange, 5 – knife edge, 6 – O-ring groove, 7 – bolt holes, 8 – thermocouple port, 9 – gas inlet, 10 – gas outlet [11].
Johnson et al. [12] have described two cells (Figures 8 and 9) for IR emission spectroscopy (IRES) studies of metal oxide catalysts.
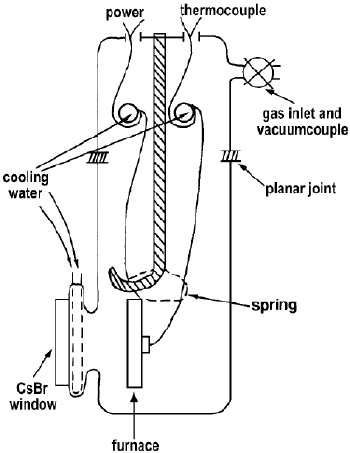
Figure 8. IRES cell made out of Pyrex glass (or quartz) and with furnace of stainless steel [12].
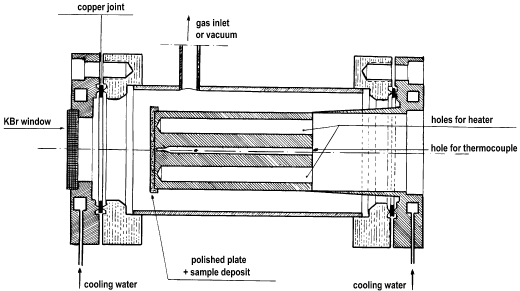
Figure 9. IRES cell made of stainless steel [12].
Arakawa et al. [13] have reported details of a novel high-pressure FT-IR spectroscopic system combined with a specially designed in situ IR cell for studying heterogeneous catalytic reactions (Figure 10).
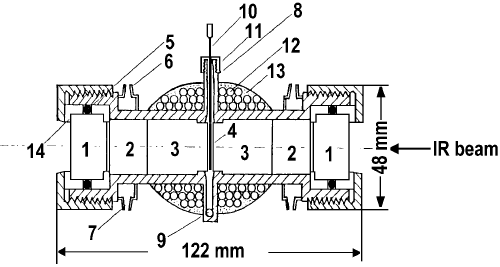
Figure 10. In situ high-pressure and temperature IR cell: 1 – CaF2 window, 2, 3 – KBr rod, 4- catalyst disk, 5 – Viton O-ring, 6 – cooling water inlet, 7 – cooling water outlet, 8 – gas inlet, 9 – gas outlet, 10 – thermocouple, 11 – Swagelok fitting, 12 – heater, 13 – asbestos molding, 14 – Teflon window holder [13].
Prokopowicz et al. [14] have presented a design of a transmission IR cell for the high-temperature study of transient adsorption and reaction in a flow system (Figure 11).
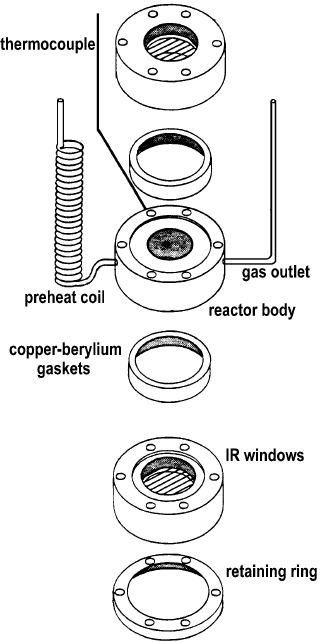
Figure 11. Exploded view of the FT-IR cell/reactor [14].
Larkins and Nordin [15] have described a high-temperature IR cell for in situ studies of the catalysts for methane oxidative dehydrogenation (Figure 12).
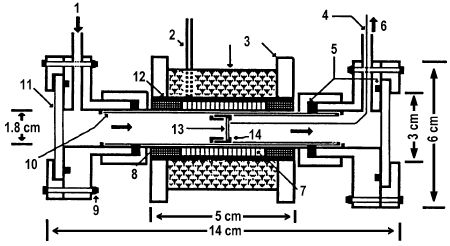
Figure 12. The high-temperature IR cell: 1 – gas inlet, 2 – electrical connection, 3 – insulating material, 4 – Viton O-ring, 5 – thermocouple, 6 – gas outlet, 7 – heating element, 8 – ceramic, 9 – nut and bolt, 10 – Vycor tubing, 11 – CaF2 window, 12 – Al2O3 tubing, 13 – sample, 14 – stainless steel sample holder [15].
Echoufi and Gelin [16] have applied IR using the cell shown in Figure 13 to measure CO physisorption on zeolites.
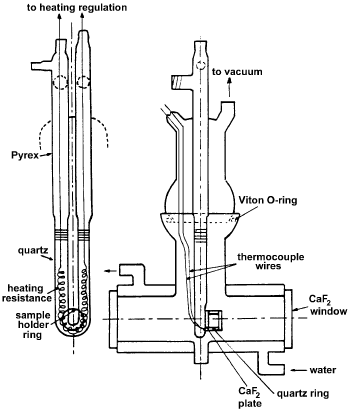
Figure 13. Schematic presentation of the IR cell [16].
Van Neer et al. [17] have reported on a reactor for IR experiments in a flow system (Figure 14).
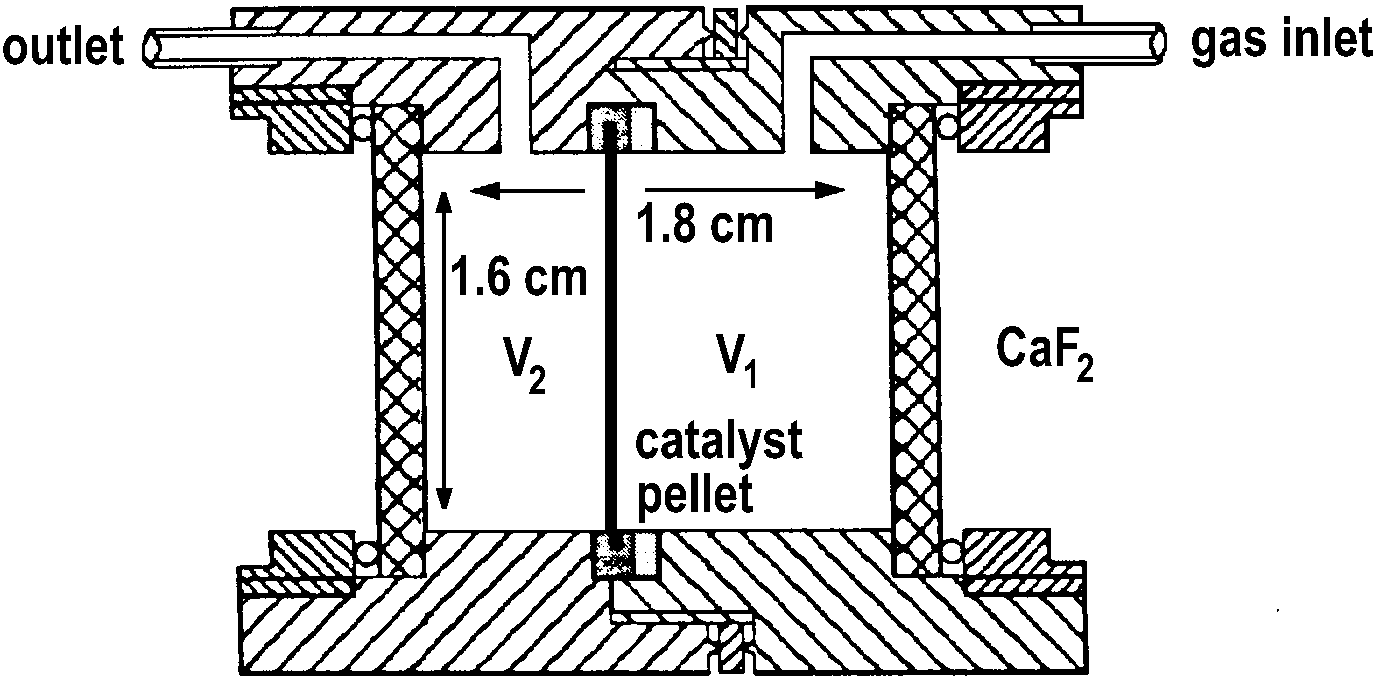
Figure 14. FT-IR flow cell (the catalyst pellet is permeable for gases) [17].
Chafik et al. [18] have applied their high-temperature IR cell for transient experiments (Figure 15).
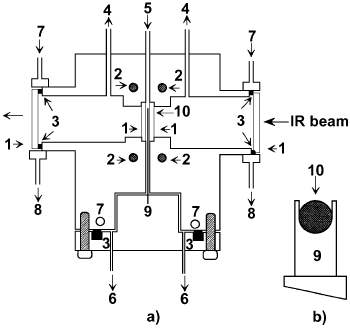
Figure 15. Design of the high temperature IR cell for transient experiments: a) body of the cell, b) sample holder; 1 – CaF2windows, 2 – catridge heaters, 3 – Viton O-rings, 4 – vacuum line, 5 – gas inlet, 6 – gas outlet, 7 – cooling water inlet, 8 – cooling water outlet, 9 – sample holder, 10 – disk of catalyst [18].
Kardash et al. [19] have reported the design and performance of an insulated IR spectro-electrochemical cell that is capable of operation at temperatures other than ambient (Figure 16).
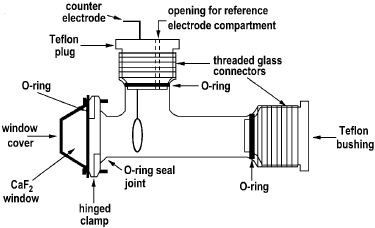
Figure 16. Cell assembly for IR spectroelectrochemical measurements at above ambient temperatures [19].
Burcham et al. [20,21] have utilized a “fixed-bed” IR cell for catalytic studies (Figure 17).
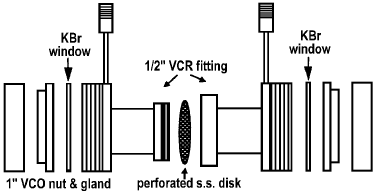
Figure 17. Schematic diagram of the “fixed-bed” IR cell [20,21]

Figure 17a. Example of glass reactor for infrared experiments (due to kindness of Prof. P. Kirszensztejn from the Faculty of Chemistry Adam Mickiewicz University in Poznan, Poland
Figures 18-21 show examples of commercial equipment. However, the last one (Figure 21) is not an attachment which can be used for in situ studies. It is the only commercially available detector for FT-IR/photo acoustic spectroscopy measurements, and can be applied for non transparent samples (e.g., carbon deposits, metal catalyst precursors, etc.).

Figure 18. In situ high-low pressure IR reactor [22].

Figure 18a. In situ high-low pressure IR reactor before the experiments (not fully connected).

Figure 18b. In situ high-low pressure IR reactor and the Bio-Rad FT-IR spectrometer (Faculty of Chemistry, University of Maria Curie-Sklodowska, Lublin, Poland).

Figure 19. High temperature IR cell [23].


(a) (b)
Figure 19a. Temperature-controlled and high-pressure demountable liquid cells (a and b, respectively [23].

Figure 19b. High-temperature and high-pressure cell [24]

Figure 20. MTEC model 300 photoacoustic detector [25].

Figure 20a. MTEC 300 PAS detector before the experiments (Faculty of Chemistry, University of Maria Curie-Sklodowska, Lublin, Poland).
There is no doubt that there is an increasing interest in the application of infrared in catalysis. Two of the classical IR techniques are still the most popular – transmission and diffuse reflectance. This is largely connected to the difficulties encountered with in situ studies, which nevertheless are of increasing significance. Moreover, monitoring for the presence and behaviour of adsorbed molecules on metal surfaces during heterogeneous catalytic reactions is of central importance for elucidating reaction mechanisms.
Abstract
Infrared spectroscopy undoubtedly represents one of the most important tools in catalysis research. A variety of IR techniques has been and can be used in order to obtain information on the surface chemistry of different solids. Special meaning applies to investigations carried out under reaction conditions. In principle for in situmeasurements, all forms of IR spectroscopy are suitable. For most practical experimental reasons, however, the transmission-absorption and diffuse reflectance techniques are best suited. This is more related to the design of cells that are to be used as reactor, than with the principal problems of the other techniques. Material presented here is partially based on the review recently published [1].
References
- J. Ryczkowski, Catal. Today, 68 (2001) 263 and references cited therein.
- B.M. Weckhuysen, Chem. Commun., (2002) 97.
- http://www.harricksci.com/infoserver/Sampling%20Techniques/techniques.cfm
- J.B. Peri, in Catalysis (J.R. Anderson and M. Boudart, Eds.), Vol. 5, Springer Verlag, Berlin, 1984, pp 172-220.
- J.E. Mapes and R.P. Eischens, J. Phys. Chem., 58 (1954) 1059.
- R.P. Eischens, S.A. Francis and W.A. Pliskin, J. Phys. Chem., 60 (1956) 194.
- J.B. Peri, R.B. Hannan, J. Phys. Chem., 64 (1962) 1526.
- R.G. Greenler, J. Chem. Phys., 37 (1962) 2094.
- P.R. Ryason, Rev. Sci. Instrum., 44 (1973) 772.
- Y. Amenomiya, Appl. Spectrosc., 32 (1978) 484.
- R.F. Hicks, C.S. Kellner, B.J. Savatsky, W.C. Hecker, A.T. Bell, J. Catal., 71(1981) 216.
- B. Jonson, B. Rebenstorf, R. Larsson, M. Primet, Appl. Spectrosc., 40 (1986) 798.
- H. Arakawa, T. Fukushima, M. Ichikawa, Appl. Spectrosc., 40 (1986) 884.
- R.A. Prokopowicz, P.L. Silveston, F.L. Baudais, D.E. Irish, R.R. Hudgins, Appl. Spectrosc., 42 (1988) 385.
- F.P. Larkins, M.R. Nordin, Appl. Spectrosc., 42 (1988) 906.
- N. Echoufi, P. Gelin, J. Chem. Soc. Faraday Trans., 88 (1992) 1067.
- F.J.R. van Neer, B. van der Linden, A. Blik, Catal. Today, 38 (1997) 115.
- T. Chafik, O. Dulaurent, J. L. Gass, D. Bianchi, J. Catal., 179 (1998) 503.
- D. Kardash, J. Huang, C. Korzeniewski, J. Electroanal. Chem., 476 (1999) 95.
- L.J. Burcham, I.E. Wachs, Catal. Today, 49 (1999) 467.
- L.J. Burcham, M. Badlani, I.E. Wachs, J. Catal., 203 (2001) 104.
- http://www.harricksci.com/accessories/H_hightemperaturecell.cfm?R=0&A=N
- http://www.in-situresearch.com/Prods/prod.html
- http://www.specac.ltd.uk
- http://www.mtecpas.com
REF: J. Ryczkowski, Int. J. Vib. Spect., [www.irdg.org/ijvs] 6, 2, 2 (2002)