Raman spectra of gases
Editor
IJVS always tries to cover all the available methods of measuring the vibrational spectra of compounds so we must, of course, consider Raman.
If you can remember back to your student days, you will remember that if a vibration involves a change in dipole as it progresses, then absorption will occur at the vibrational frequency and the vibration will give rise to a characteristic absorption based in the IR. If there is a change in polarizability, a Raman scattering band can be observed. So – different vibrations will give rise to infrared and Raman spectra and hence the two spectra of a given compounds will certainly not look the same. These comments apply equally to solids, liquids and gases so in principle gas phase Raman spectra can be useful.
In last edition’s article [Volume 5, Edition 3], I pointed out that gases rotate and vibrate generating Rotation and Rotation Vibration spectra. Similar effects are seen in Raman spectroscopy.
I won’t bore you with the selection rules, but a clear series of equi-spaced Raman lines due to rotation can be seen. The bands lie very close to the exciting line because the energy of rotations is very low. Further, the lines are very close together unless the molecules have very low moments of inertia.
To give you a ‘feel’, the rotational lines in nitrogen, oxygen and chlorine are spaced apart by 8, 14# and 2cm-1 respectively. Large molecules will have separations so close that experimental methods capable of separating them are hard to come by and certainly not of much interest to us.
# The moments of inertia of N2O2 and Cl2 must go in the order N2<O2<Cl2, thus the line separation of O2 is peculiarly large. The reason is that every other rotational line is missing whereas all lines show up in N2+Cl2.
Turning now to rotation vibration spectra – sharp equi-spaced rotation-vibration Raman bands surround the simple vibrational transition just as they do in the infrared. However, there are several differences: –
Because of the difference between the selection rules for infrared and Raman spectra, the rotational wings are twice as broad in the Raman as they are in the infrared, seemingly a useful experimental advantage, BUT the Q branch in the Raman is almost always much more intense than the rotation-vibration bands. Incidentally, the pure Rotational bands are usually more intense than the Q branches of the vibration-rotation spectra.
So, we have a distinct set of differences between the IR and Raman spectra of vapours and gases
IR → Rotations give bands below the lower frequency limits of most FTIRS.
Rotation-Vibration spectra come in several band shapes and if the vibrating molecules are small, the P&R branches may well appear as clearly resolved sharp bands.
Raman → Because one can scan spectra very close to the existing line. Rotation spectra can be easily recorded and are relatively intense.
Rotation-Vibration spectra are not often recorded because the Q branch is so much more intense than the vibrational /rotation wings, so we see pure Rotational spectra at very low energies and essentially pure vibrational spectra at larger shifts.
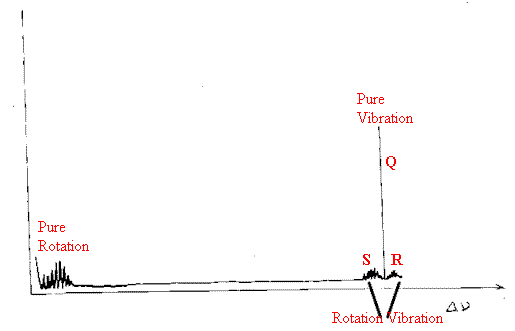
Figure A.
Thus, Raman looks good from an analytical point-of-view. However, there is a problem – Raman spectra are always weak and in gases – really weak. As a result, relatively little Raman is done in the gas phase. Raman is sometimes used because of its ability to see both rotations and vibrations of homonuclear diatomics – something that absorption techniques simply cannot do. Thus, people have used Raman to monitor oxygen or nitrogen levels. Similarly, chlorine or bromine. Good examples where homopolar diatomics are involved are headspace analysis in the pharmaceutical industry [tablets are sometimes packed under inert atmospheres. Leakage of O2 can be disastrous to shelf life. Raman provides a non invasive analytical test]. Bubbles occur all over the place and especially in minerals. The gases inside the bubbles are routinely analysed by Raman using petrographic sections and the Raman microscope. The appearance of the spectrum changes with pressure. Increasing pressure broadens out the otherwise needle sharp bands. The spectrum of a bubble in a mineral can therefore indicate both the contents and the pressure.
The gases used in anaesthetics and the composition of exhaled gases can be crucially important. Although many techniques exist for this type of analysis, Raman is proving its value especially as the cost of Raman modules reduces. One further advantage Raman has over infrared is its ability to assay a sample (including a gas or vapour) from one side.
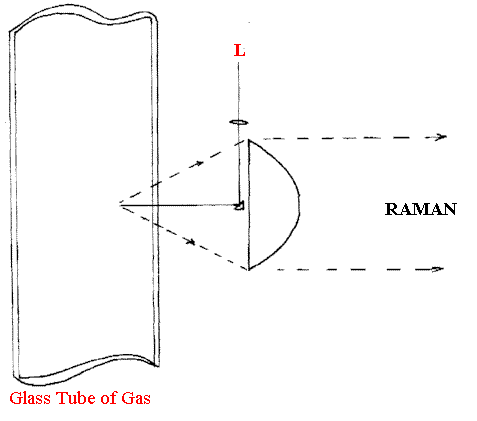
Figure B.
This feature can be invaluable – for example, people have remotely examined the composition of the gases leaving smokestacks. See Figure C.
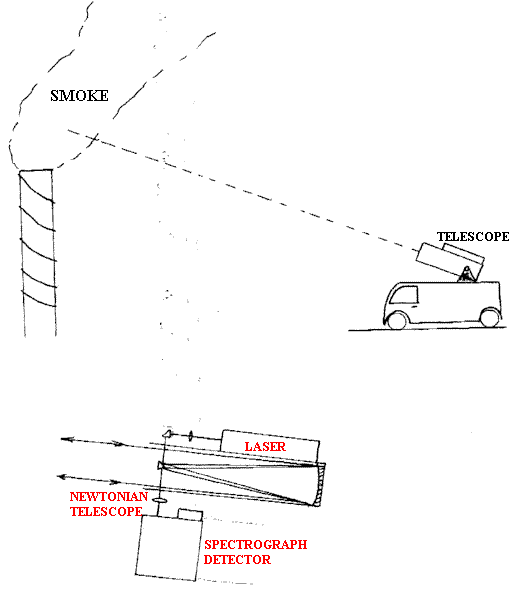
Figure C.
If used in an enforcement regime, owners of chimneys are unlikely to be very helpful, so setting up an infrared absorption system or taking samples in gas ampoules may not be easy, but Remote Raman can be applied from outside the factory fence. Another area where one-sided analysis is essential is in the study of high and variable temperature flows of gas. The temperature gradients deflect the beam so transmission IR is unattractive. If the Raman experiment is done confocally, the gradients are no longer a nuisance. See Figure D.
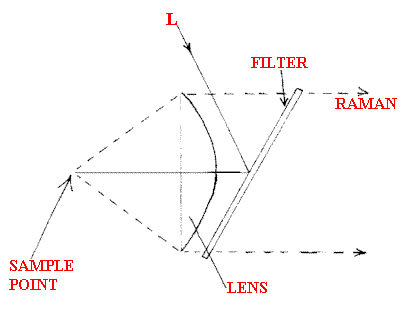
Figure D.
Just such an area where analytical results are needed and can be obtained using Raman is in the analysis of the gases leaving aircraft jet engines. The gas stream can be moving at supersonic speeds hence temperature and sample probes will generate shock waves, interfere with the gas streams and provide suspect results. Raman being non intrusive is just the trick. I am told that using Raman data the performance of each combustion ‘can’ of a jet engine can be tuned and the engine performance optimised more rapidly and easily then before when only visible observation could be used. Analysis of the gases in an exhaust stream is one problem but the temperature is another piece of data essential to the engineers. As I have said above, temperature sensors generate shock waves and are unattractive, but the Raman spectrum can indicate the temperature directly i.e. composition and temperature come out of the one experiment. The principle is a very old one. The energy level diagram for the Raman process is shown below –
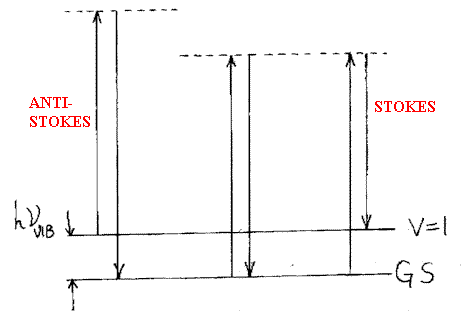
hence the intensity of the bands depends amongst other things on the population of the ground and excited states. The latter depends in turn on the temperature. Hence, if we identify a Stokes (red shifted) and Anti-Stokes (blue shifted) pair of bands the relative intensity will be related to the population and hence
e –uVIB/kT
Crudely –
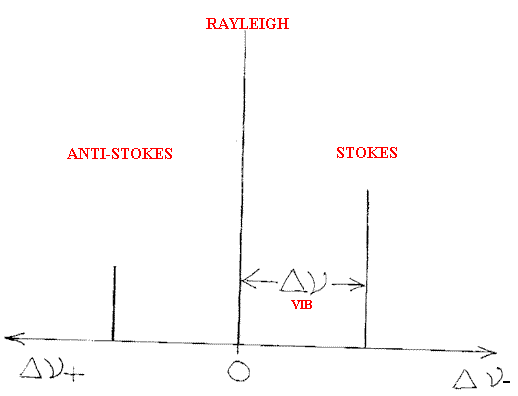
Another area where Raman can in principle provide unique data is in the study of flames.
In a stable flame a cold combustible mixture of gases moves in one direction and the flame front moves in the opposite. If stable, the flame front will be fixed. The flame front (really a lamella zone) is very thin, the thickness increasing as pressure is reduced. See Figure E.
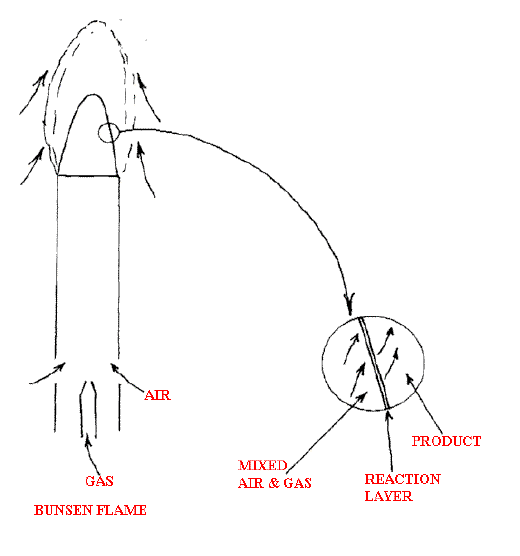
Figure E.
For years combustion scientists have wanted to analyse the gases and temperatures throughout flames, but there are problems. Reliable temperatures are hard to measure because the sensor interferes with the process. IR emission thermometers are not satisfactory because the flame is always three-dimensional and complex. Sampling is problematic because the gases will inevitably react inside the sampling tube. Raman suffers from none of these disadvantages. The only problem is the emission from the flame -–emission bound to be much more intense than the Raman scatter. This problem has been solved. If the laser is periodically interrupted with a chopper, the Raman scatter will also be chopped. Flame emission on the other hand will be continuous so if the output is fed through an electronic device capable of looking for the chopped rather than the continuous signal (a phase sensitive detector (PSD)) the Raman scatter can be picked out. In Figure F. I give some examples including a spectrum of the gases in a domestic gas, air flame. The Raman spectrum shown in Figure F is weak but perfectly useable. Oddly the pioneering measurements have not been developed and exploited although there really is no reason why they shouldn’t be. Raman can be useful in all the investigation of flames, plasmas, and combustion inside furnaces or engines.
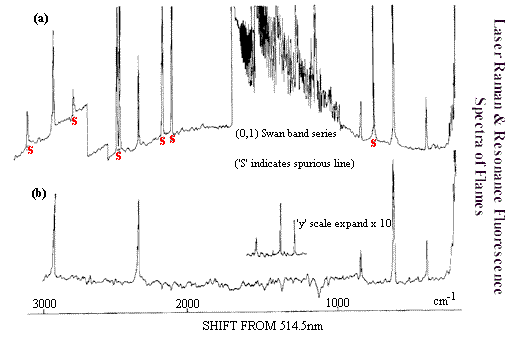
Figure F. Top spectrum – total emission & scatter from flame.
Bottom spectra – using phase sensitive detection only the
Raman bands are seen. At very low frequencies – N2 & O2
rotational bands below 1000cm-1 rotational bands of H2.
Above vibrational bands due to N2, O2, CO2 & CH4.
Taken from P.J.Hendra, C.J. Vear & J.J. Macfarlane, Advances in Raman Spectroscopy I, Heyden & Son Ltd London. Proceedings of IIIrd International Conference on Raman Spectrscopy Reims, September 1972, Paper 30.
Perhaps one reason why conventional Raman has not been exploited in this field was the development of coherent antistokes Raman Spectroscopy (CARS). This technique involves the illumination of the sample (and gases work really well) with two laser beams coincidentally. It turns out that the early experiments looked really interesting but the developments needed to make CARS routine just didn’t happen. Perhaps the instrument manufacturers couldn’t see a big enough market. It’s a pity
[One day we should have an edition devoted to this multiphoton phenomenon and also include coherent Raman scattering, CARS and Raman absorption.]
So Raman of gases is difficult, but well worthwhile in specific cases. Much more on the technique can be found by consulting the Handbook of Raman Spectroscopy, Eds I.R. Lewis & H.G.M. Edwards, Marcel Dekker NY 2001. See Chapter 9 (p307) by H.W. Schrötter and Chapter 23 (p919) by I.R. Lewis.
REF: P.J.Hendra, Int. J. Vib. Spect., [www.irdg.org/ijvs] 5, 4, 3 (2001)