Determination of explosion and decomposition gases with IR spectroscopy
Anton Hammerl,a Gerhard Holl,b
Thomas M. Klapötke*,a and Gunnar Spießa
a Department of Chemistry,
University of Munich,
Butenandtstr. 5-13 (D),
D-81377 Munich, Germany,
fax ++49-(0)89-21807492,
e-mail: Thomas.M.Klapoetke [at] cup.uni-muenchen.de
b Wehrwissenschaftliches Institut für Werk-,
Explosiv- und Betriebsstoffe,
Grosses Cent,
D-53913 Swisttal (Germany)
Abstract
The explosion of several nitrogen rich explosives was initiated by electrical resistance heating. The explosion gases were determined by IR spectroscopy and mass spectrometry.
Introduction
Polynitrogen and nitrogen rich materials have high energy densities and are, therefore, promising high energetic materials for use as explosives and propellants [1, 2]. We are investigating hydrazinium azide derivatives [3, 4], methylated hydrazinium nitrates and 5,5´-azotetrazolate salts [6, 7]for use as high nitrogen highly energetic materials.
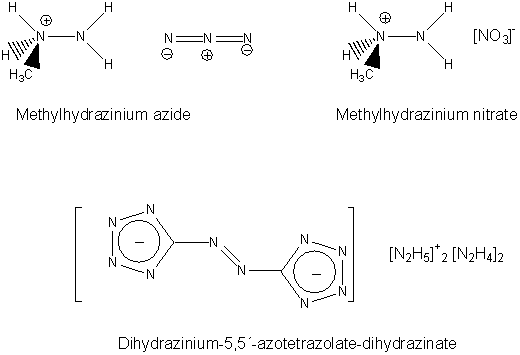
Hydrazinium azide derivatives and azotetrazolate salts have both negative oxygen balances, which means that the molecule needs additional oxygen for a complete combustion to CO2 and N2. In commercial explosives, e.g. nitroglycerin, the oxygen balance is often negative. The molecule contains more oxygen than needed for a complete combustion. As a result, NOx are formed during the explosion. To avoid this, materials with negative oxygen balance are added to form explosive mixtures.
Therefore, the explosion gases of high energetic materials have to be determined. Here we report on a strategy for the detection of explosion gases combining infrared spectroscopy and mass spectrometry. The processing of the resulting spectra is shown for methylhydrazinium azide, methylhydrazinium nitrate and dihydrazinium-5,5´-azotetrazolate-dihydrazinate.
Experimental
To detect the explosion gases, the explosion of a high energetic material has to be initiated in a gas free environment. A sample of the material was placed in a steel bomb. The sample is in contact with a spiral wire. Then the steel bomb was evacuated. An electrical current from a lead akkumulator is applied to the wire. The red hot wire initiates an explosion.

Picture1: Steel bomb with sample container and spiral wire.
The bomb containing the explosion gases can then be connected to a mass spectrometer via a Whitey connection.

Picture 2: Steel bomb attached to mass spectrometer.
To record an infrared spectrum of the explosion gases, the gases have to be transferred to gas cell that has infrared transparent windows. We accomplished this by condensating the gases in the IR gas cell on a metal vacuum line with Whitey connections.

Picture 3: IR gas cell at steel vacuum line.
The IR spectra were recorded on a Perkin Elmer Spectrum ONE FT-IR spectrometer, the mass spectra on a Balzers QMG 420 mass spectrometer.
Determination of explosion gases
The process of identifying the explosion gases is demonstrated for several compounds. It is important to combine the information obtained from the IR and MS measurements to completely identify the explosion gases. Spectra of compounds in the gas phase for comparison were obtained from webbook.nist.gov.
Homonuclear, diatomar gases show no absorptions in the infrared spectra, only in the raman spectra. Therefore, these gases can only be identified in the MS spectra.
Methyl hydrazinium azide
Methyl hydrazinium azide explodes to form dinitrogen, dihydrogen, methane and ammonia. Small amounts of HN3 were also present.
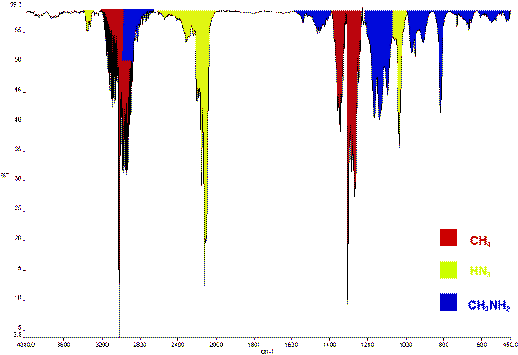
Figure 1: IR gas spectrum of explosion products
of methylhydrazinium azide.
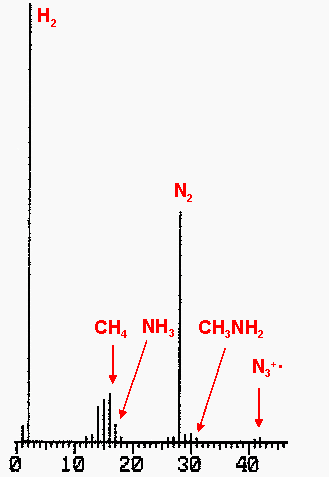
Figure 2: Mass spectrum of explosion gases
of methylhydrazinium azide.
Dihydrazinium-5,5´-azotetrazolate-dihydrazinate
Dihydrazinium-5,5´-azotetrazolate-dihydrazinate explodes to form dinitrogen, dihydrogen, methane and ammonia. The infrared spectrums shows only weak absorbtions for methane and ammonia. Dinitrogen and dihydrogen are detected in the mass spectra.
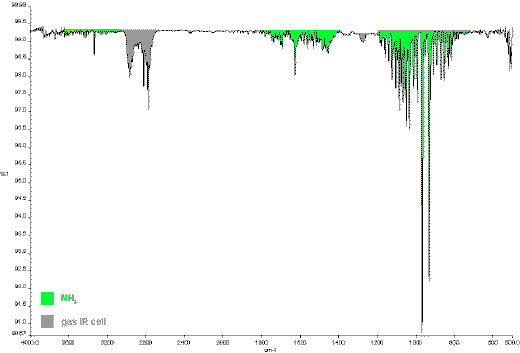
Figure 3: IR gas spectrum of explosion gases of
dihydrazinium 5,5´-azotetrazolate dihydrazinate.
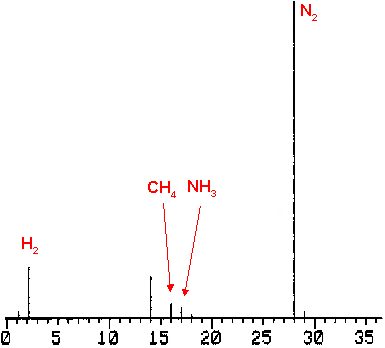
Figure 4: Mass spectrum of explosion gases of br /> dihydrazinium 5,5´-azotetrazolate dihydrazinate.
Methylhydrazinium nitrate
Methylhydrazinium nitrate is less explosive than the first two compounds. Methylhydrazinium nitrate is found as a solid residue in rocket engines with methylhydrazine and N2O4 as propellants. Methylhydrazinium nitrate does not explode in the steel bomb, it decomposes. The infrared spectrum shows absorptions from many compounds that are produced during an incomplete decomposition. This makes the interpretation of this decomposition difficult. The bands in the infrared spectrum are assigned to NO, N2O, CO, CO2, CH3NH2, CH4, NH3 and NO2 . The mass spectrum shows that N2, H2O and CH3N3 are also produced during the decomposition.
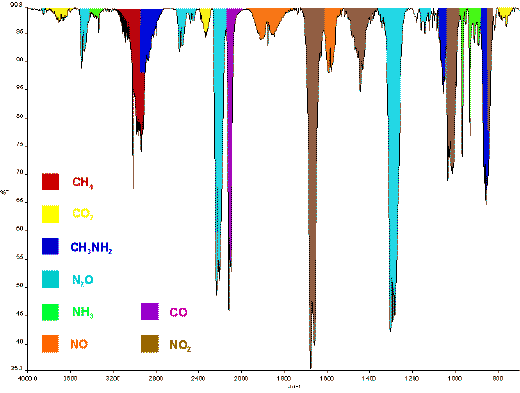
Figure 5: IR gas spectrum of decomposition
products of methylhydrazinium nitrate.
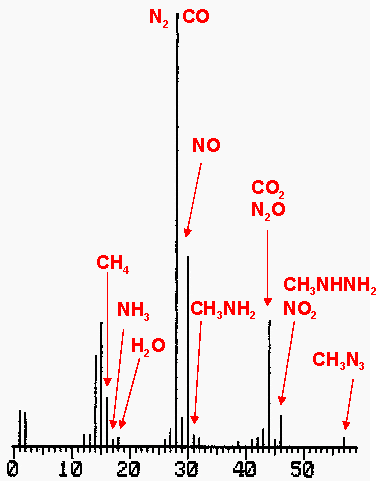
Figure 6: Mass spectrum of decomposition
gases from methylhydrazinium nitrate.
Conclusion
It is possible to analyse the explosion gases by IR spectroscopy and mass spectrometry. However, a combination of IR and mass spectra is necessary to identify all gaseous products because diatomic, homonuclear gases like H2 and N2 do not show absorptions in the IR spectra. We have shown that the explosions of methylhydrazinium azide (H2, N2, CH4, NH3 and CH3NH2) and especially hydrazinium 5,5´-azotetrazolate dihydrazinate (N2, H2, NH3, CH4) produce only low mass explosion products with dihydrogen and dinitrogen being the main products.
In decomposition reactions, like the decomposition of methylhydrazinium nitrate, more products are formed. Therefore, it is more difficult to analyse the gaseous decomposition products. The decomposition of methylhydrazinium nitrate gives a mixture of nitrogen (NO, NO2, N2O) and carbon oxides (CO, CO2) as well as decomposition products of the methylhydrazinium molecule (CH3NHNH2, CH3NH2, CH4, NH3). Remarkably, methylazide is also formed during the decomposition.
Acknowledgment
Financial support of this work by the University of Munich (LMU), the Deutsche Forschungsgemeinschaft (DFG, KL 636/7-1) and the German Federal Office of Defense Technology and Procurement (BWB) is gratefully acknowledged.
References
- K. O. Christe, W. W. Wilson, J. A. Sheehy, J. A. Boatz, Angew. Chem.1999, 111, 2112-2117, Angew. Chem. Int. Ed. 1999, 38, 2004-2009.
- D. E. Chavez, M. A. Hiskey, R. D. Gilardi, Angew. Chem. 2000, 112, 1861; Angew. Chem. Int. Ed. 2000 39, 1791.
- A. Hammerl, G. Holl, K. Hübler, T. M. Klapötke, P. Mayer, Eur. J. Inorg. Chem. 2000, 755-760.
- A. Hammerl, G. Holl, M. Kaiser, T. M. Klapötke, P. Mayer, H. Nöth, M. Warchhold, Z. Anorg. Allg. Chem., in press.
- O. de Bonn, A. Hammerl, T. M. Klapötke, P. Mayer, H. Piotrowski, H. Zewen, Z. Anorg. Allg. Chem., in press.
- A. Hammerl, G. Holl, M. Kaiser, T. M. Klapötke, H. Nöth, U. Ticmanis, M. Warchhold, Inorg. Chem., in press.
- A. Hammerl, G. Holl, M. Kaiser, T. M. Klapötke, P. Mayer, H. Nöth, H. Piotrowski, M. Suter, Z. Naturforsch. B, submitted.
Received 8th June 2001, received in revised format 25th June,
accepted 25th June 2001.
REF: Anton Hammerl, Gerhard Holl, Thomas M. Klapötke and Gunnar Spieß Int. J. Vib. Spect., [www.irdg.org/ijvs] 5, 3, 6 (2001)