The problem of sample heating in F-T Raman Spectroscopy
The Editor
Recently, Bowie, Chase & Griffiths [1] have contributed feature articles in Applied Spectroscopy describing sampling in Raman Spectroscopy. The articles are to be recommended because they survey in a most readable form, the advantages and disadvantages associated with Raman measurements.
Quite correctly, Chase & Griffiths point out that one of the snags with Raman spectroscopy and particularly in the F-T technique is sample heating, but they offer very little information about controlling it. So I thought I would help!
F-T Raman spectroscopy has made a real impact on analytical chemistry. The method relies on illuminating the sample with a near infrared laser and processing the scattered light on a good quality FTIR bench. Most of the leading FTIR manufacturers offer a ‘Raman Accessory’ and sales have been brisk for at least 10 years. With a Raman set-up on an FTIR you can record the two IR and Raman complimentary spectra on your sample. Raman has, in the past, failed to gain widespread acceptance within the analytical community because sample colour and fluorescence have made it almost impossible to use it as a ROUTINE tool. Infrared is (or rather was) much more versatile in that the vast majority of a ‘basket’ of samples will give an infrared spectrum of sorts. In Raman, this wasdefinitely not the case. When Chase demonstrated that NIR/FTR was feasible back in the mid 1980’s [2], this restriction was largely removed – probably 85% of a random bunch of samples will give an F-T Raman spectrum. However, one major cause of failure is sample heating.
In F-T Raman, laser powers of -w are typical. The laser is focussed into a patch on the sample around 0.5mm in diameter hence the brightness of illumination is
0.52/ p(1/40)2~300w cm-2, ample to heat any sample which absorbs the laser radiation even weakly. If the sample absorbs the laser radiation efficiently it will be ‘burned to a cinder’!
Strictly, the brightness and any absorption are not in themselves a problem – the absorption of the laser is an energy term, which becomes serious when multiplied by time. Put another way, the sample heats at a rate dependent on the brightness of illumination and the absorption coefficient at the laser wavelength [1.064i]. As it turns out, a huge number of samples do indeed have a very small but significant coefficient near 1 i (particularly samples that are wet, have water of crystallisation, contain OH and or NH groups), so some degree of heating is very widespread. In most cases, this causes little or no problem, but it can be a headache in a low melting material. An example is shown in Figure 1, the sample has melted but the analyst wanted the spectrum of the solid!
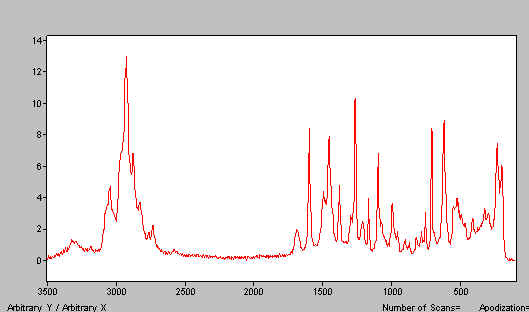
Figure 1. Lidocaine – this spectrum is that of the melt
Quite obviously, the rate of heating and of course, heat loss vary from sample to sample and due to differences in design will vary from instrument to instrument but in my experience a 5K rise in temperature is routine and in heavy absorbers several hundreds of Kelvin definitely occur. To reduce the problem, the tricks are really three-fold.
- Arrange for a short efficient thermal path for the heat energy to be dissipated.
- Cool the sample so that the problem simply disappears and,
- In effect, increase the area of illumination so that the brightness is reduced or is reduced over time.
Let us consider each in turn.
Heat dissipation
The principle here is simple – dilute the sample with an efficient conductor of heat and provide a pathway for the energy to dissipate and hey presto – the problem is solved – maybe! The alkali halides have thermal conductivities as high as metals and so two Raman solutions have been reported.
1) use a near infrared type KBr disk and 2) try a high concentration liquid paraffin mull squeezed between KBr flats. The KBr disks recommended by reporters tend to be thicker and more concentrated than for IR absorption measurements and similarly the thickness of a mull for Raman work is certainly greater than is typical of IR usage. Both methods work well and FT Raman spectra of graphite – and you can’t get much blacker than that – have been reported. A good convenient source of further information will be found in IJVS [3]. An article about mulls follows this one.
Quite obviously, this is attractive because with a little bit of forethought the IR and Raman spectra can be recorded on the same sample very easily. However, this method does have its limitations – use too much laser power and an unacceptable level of heating will still happen, alternatively wait forever for the results! The dark colour usual in these cases (vide graphite) reduces the optical efficiency of the overall process thus long scan times are inevitable.
Sample cooling and cold cells
If the sample is heated by the laser, one almost trivial way of controlling the problem is to cool it to cryogenic temperatures. The sample will then tolerate a rise in temperature of a couple of hundred K and still be far from overheated. A temperature rise of 150 or 200K is unlikely to precipitate a disaster. So we need a cryocell – what is available?
This subject has been covered before in IJVS [4], but at the risk of boring you, I will go over some of the material again. There are in effect, three types of cryogenic cells – ‘cold finger’, streaming cold gas and ‘transfer gas cells’. All are drawn in Figure 2.
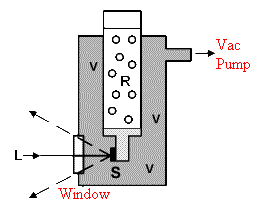
Figure 2a. Cold Finger Cell
V= Evacuated spaces
S= Sample, L=Laser input
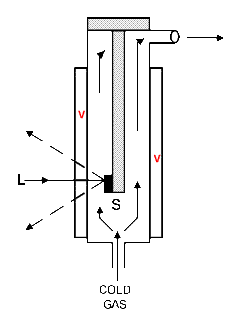
Figure 2b. Steaming Gas Cell
V= Evacuated spaces
S= Sample, L=Laser input
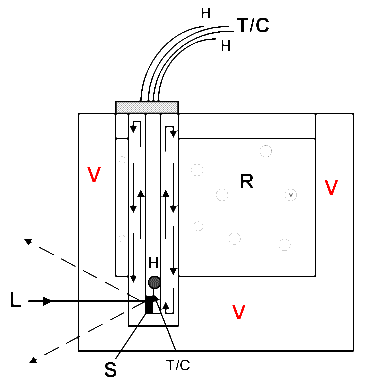
Figure 2c. Transfer Gas cell
V= Evacuated spaces (sealed and permanent in Figures 2b and 2c.)
S= Sample, T/C= Thermocouple, H=Heater, R=Refrigerant, L=Laser input
The sample and cold finger are encased in an evacuated zone – again not really appropriate for our purpose because continuous pumping or at least semi-continuous evacuation is required. So we move on to the streaming gas cells. As you will see in Figure 2b, the sample simply dangles in a flow of nitrogen boiling off the liquid. These systems are simple, work well but it is hard to precisely control the temperature of the sample. You can increase the temperature of reducing the flow rate and vice versa, but its all a bit hit or miss.The cold finger design of cell requires that the sample is clamped to a refrigerated surface and is in excellent thermal contact with it (particularly if the whole ides is to draw away the laser energy to minimise heating). In electronics, thermal transmission is maximised by using a special paste squeezed between solid state devices and heat sinks but this solution is not usually applicable in Raman because the paste has a spectrum (and fluorescence) all its own.
The third alternative, is the ‘transfer gas’ cell. As you will see in Figure 2c a gentle and continuous flow transfers heat from the sample to the refrigerant. The latter need not involve boiling gases – solid CO2/acetone or even ice/NaCl are just fine. The sample holder can contain a tiny heater and thermocouple and these can be used to accurately control the temperature at a selected value. The sample sits in a cold gas cell, not a vacuum so thermal conduction of the laser energy away from the sample is maximised and also samples can be changed rapidly and easily (no vacuum to release and re-pump).
There is a further advantage with the transfer gas cell appropriate only to F-T Raman. Manufacturers of our instruments almost invariably seem to think we don’t need any space around the sample zone – “small is beautiful” must be their collective motto! Further, the standard arrangements of the sample chamber (which is always interlocked with the laser) make it advantageous to minimise the tubes and cables connecting the cell to the outside laboratory. Now, the cold finger cells need vacuum hoses, the streaming gas cells require a large and insulated gas transfer tube. Both systems are hard to accommodate in a small sample area with a closed lid. Transfer gas cells are made from glass, the Dewar vessels sealed and they require only thermocouple and fine heater cables to pass to an external power supply. An example of a simple transfer gas cell will be found in Figure 3. I could say more about these cells and describe more sophisticated designs which have recently appeared in IJVS, but I will leave the matter there. For new designs see IJVS [5].

Figure 3. The Ventacon Cold Cell in a Nicolet Raman System
Increasing the area of illumination
Two approaches are possible here – increase the patch illuminated by the laser, thus lowering the brightness and/or move the sample under the laser beam so that a larger area is illuminated and time is given for the heat to dissipate.
Let’s consider the simple ruse of de-focussing the laser beam. There is a problem. The instrument “looks” at a patch on the sample, which is a virtual de-magnified image of the entrance Jacquinot stop. See Figure 4. To put in some fairly typical figures – at analytical levels of resolution (2-4cm-1) manufacturers use a ‘J’ stop size around 6mm. Most magnify the sample at the ‘J’ stop plane by about 6x so the instrument sees a patch only about 1mm in diameter at the sample. This is why the laser is focussed down to a spot around -mm in diameter – it drops easily into the viewed area [BioRad focus least of the suppliers]. Enlargement of the illuminated patch is permissible but only within the viewed area. So, the opportunity to reduce burning this way is very restricted and in my personal opinion not worth the trouble. To do the job, you must change the manufacturer’s laser focussing lens and then carefully align the illuminated and viewed patches. OK, if you are good at “Do-It-Yourself”, but otherwise fraught with problems over manufacturer’s warranty, servicing etc!
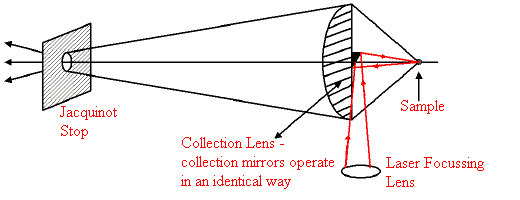
Figure 4.
The alternative is sample movement (usually rotation). The idea here is to illuminate the sample as it moves so that each part is heated by the laser for only a very short instant and it then given time to cool down before it is heated again. Simple rotation cells have been used for some years and Nicolet owners have bought considerable numbers. An example is shown in Figure 5. To prove that the method works, you will see in Figure 6, a spectrum of solid lidocaine. This very low melting material is a liquid under a stationary beam (Figure1) but not if it is kept moving.
 Figure 5. The Ventacon Sample Rotator and Battery Pack
Figure 5. The Ventacon Sample Rotator and Battery Pack
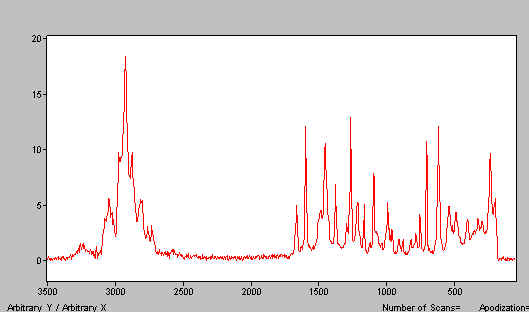
Figure 6. Lidocaine – spectrum recorded exactly as Figure 1,
but the sample was rotated. Both spectra were obtained at room temperature.
You don’t have to rotate alone – Franz Langkilde at Astra Zeneca, in Mölndal in Sweden, demonstrated the value of rotation and translation occurring together and this too have been developed/. See Figure 7. Samples held in 5mm NMR tubes can be illuminated over a spiral path of height 10mm or more as they rotate, so illumination can be spread over 200mm2 very easily.

Figure 7. The Ventacon 2-axis rotator
Pharmaceutical tablets are a rather special case. Heating can indeed be a problem, but so also can inhomogeneity. Manufacturer’s make their tablets by impact compression of powder mixtures and often imprint identifying logos, numbers or letters into their surfaces. Shape also is variable, round, lozenge shaped, rounded triangular or cylindrical tablets are commonly found. The trick when examining these is to rotate and scan up and down at the same time making sure that the instrument examines a high proportion of the tablet’s surface. Heating is then minimised and further, the spectrum is that of the tablet AVERAGED over the viewed surface. Imprints are ignored. An example of such a cell is shown in Figure 8. Much more complex cells have also been developed to study vapour absorption by powders – here the sample is exposed to a vapour in a carrier gas e.g. wet air and the reaction that occurs as the vapour is absorbed is monitored. Since these processes are temperature dependent, the vapour, gas and sample are contained within a temperature controlled zone AND the sample rotates to minimise sample heating. An example can be seen in Figure 9.

Figure 8. The Ventacon Autochanger Tablet Analyser

Figure 9. Humidity Cell
Readers who are experienced in the physics of Fourier Transformation will quite correctly be nervous about the idea of moving the sample under the laser beam. The process can and will introduce noise into the spectrum. The trick is to move slowly so that the noise falls well outside the frequency domain of the F-T Raman spectrum. This subject has again been discussed in IJVS and elsewhere [6].
So, to conclude, to minimise laser heating in routine analytical laboratories, try a KBr disk or liquid paraffin mull. Alternatively buy a simple rotator and examine your samples uncontaminated in NMR tubes. If you want to be doubly sure, purchase a 2 -axis rotator – round and round and up and down. Rotation is relatively cheap, simple to use and requires little or no manipulative skill. If you want to lower the temperature use a cryocell – more expensive – more difficult to use but these accessories do often open up a range of experimental possibilities and you can look at pieces of plastic, frozen liquids, bundles of fibres, rolled up films or lumps of rubber equally easily. If a sample absorbs fairly strongly, cooling may not be enough – cooling and rotation might be the answer [I’m working on this one!]. De-focussing the laser – not a very good idea. Good luck!
References
- B.T. Bowie, D.B. Chase & P. Griffiths, Appl.Spectroscopy, 54, 164A (2000).
- D.B. Chase, Anal. Chem., 59, 881, (1987).
- Y.D. West, Int.J.Vib.Spect., [www.irdg.org/ijvs] 1, 1, 5 (1996)
- P.J. Hendra, Int. J. Vib. Spect., [www.irdg.org/ijvs] 1, 2, 6 (1996)
- M.Catlin, P.J. Hendra & M. Snow, Internet J. Vib. Spec. [www.irdg.org/ijvs] 4, 1, 4 (2000)
- A. dePaepe, J.M.Dyke, P.J. Hendra & F.W. Langkilde, Spec. Acta. 53a, 13, 2261 (1997)
Click here to view spectra for this article.
Declaration of interest The accessories shown in Figures 3, 5, 7, 8 and 9 are supplied by Ventacon Wotton Ltd and I am involved in their design and construction. I am not implying that these cells are the only ones available. It’s just laziness – I had the pictures to hand!
REF: P.J. Hendra, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 3, 2(2000)