Surface-Enhanced Raman Spectroscopy: Its Present Status
Z. Q. Tian
State Key Laboratory for Physical Chemistry of Solid Surfaces
and Department of Chemistry,
Xiamen University,
Xiamen 361005,
China
Email: zqtian [at] xmu.edu.cn
Introduction
The field of surface enhanced Raman spectroscopy was inaugurated in the mid-1970s. In 1974, Fleischmann, Hendra and McQuillan first observed the phenomenon from pyridine adsorbed on an electrochemically roughened silver electrode [1]. It was shown clearly that the good-quality signals of vibrational bands were different from the values due to pyridine in the aqueous electrolyte. In 1977, Jeanmaire and Van Duyne [2] and, independently, Albrecht and Creighton [3] concluded that the rough silver electrode produce a Raman spectrum that is at times a millionfold more intense than what was expected. This enormously strong surface Raman signal must be caused by a true enhancement of the Raman scattering efficiency itself, and has been dubbed surface enhanced Raman scattering (SERS) [4].
The discovery of SERS had impacted on surface science and vibrational spectroscopy communities because of its extremely high and unique surface sensitivity. It is well known that the most important component of any reliable and general surface analytical method is its sensitivity. Raman scattering is a second-order process, with very low cross sections. In the absence of any resonance Raman process, the differential Raman cross sections are (ds /dW )NRS£ 10-29 cm2sr-1. Hence the corresponding surface Raman intensities expected for a monolayer of adsorbates are typically less than 1 count per second (cps) when using standard Raman spectrometer systems. This intrinsically low detection sensitivity is no longer a fatal disadvantage for surface Raman spectroscopy SERS can be invoked since the signal from the adsorbate on the SERS active surface is now sufficiently strong. Such an astonishing phenomenon makes possible an in situ diagnostic probe for detailed molecular structure and orientation of surface species, widely applicable to electrochemical, biological and other ambient interfaces.
In the past two decades there have been thousands of papers and some excellent review articles and books dealing with the SERS phenomenon [4-17]. The diverse SERS systems include virtually every conceivable metal and morphology; from atomically smooth to highly rough surfaces, from thick to thin films coated on smooth and roughened substrates, from colloids to powders and even to catalysts supported on insulator granules. A large number of experimental characteristics of SERS have been discovered. It has been found that SERS differs in a number of ways from ordinary Raman spectroscopy of molecules and even from un-enhanced surface Raman spectroscopy. The key features are summarized briefly as follows:
-
SERS occurs for a very large number of molecules adsorbed on or near to the surface of metals in a variety of morphologies. The largest enhancements (with surface enhancement factor (SEF) > 106) are observed from the coinage metals silver, gold and copper, and roughened with features of submicroscopic dimensions (about 20-300 nm). The atomic scale roughness, such as certain adatoms, adclusters, steps or kinks can assist further enhancement.
-
SERS has been observed at solid/liquid, solid/gas, and solid/solid interfaces, as an interface-sensitive technique. Molecules adsorbed in the first layer on the surface show the largest enhancements. However, the enhancement also has some long-range aspect with molecules separated from the surface by tens of nanometers showing some enhancement depending upon the substrate morphology and physical environments.
-
The Raman bands from SERS active surfaces with various roughnesses (except single nanoparticles) are completely depolarized, in contrast to those taken from molecules adsorbed on atomically smooth surfaces. Overtone and combination bands are not prevalent and normally Raman inactive vibrational modes can be observed. Selection rules are relaxed resulting in the appearance of normally forbidden Raman modes in the surface spectra. SERS active surfaces also display a continuum inelastic background scattering and they are also very effective in quenching fluorescence.
-
The excitation profile (scattering intensity vs exciting frequency) deviates from the fourth-power dependence of normal Raman scattering. Both vibrational band frequencies and SERS intensities are a function of the applied electrode potential in electrochemical experiments, and the functionality may be different for different sets of vibrational modes.
These findings have stimulated extensive theoretical research on the SERS phenomenon. Among the various proposed SERS mechanisms, “electromagnetic (EM)” and “chemical effect” (or “charge transfer” (CT)) enhancements have been commonly accepted as the two main contributions to the giant surface enhancement [6,8,11,15,16]. However, there is still disagreement over the relative and absolute enhancement factors for each. This is mainly due to the fact that EM and CT models both are the critical roughness-based models. The largest EM enhancement occurs for surfaces that are rough on the nanoscale (10 – 100 nm) while the roughness from atomic to 10 nm scale creates the largest CT enhancement. When optimizing for SERS activity, most of the SERS active surfaces are very rough and look like cauliflowers, consisting of various roughness components ranging from atomic- to submicron-size. Therefore, it is very difficult to distinguish the contribution from the EM and the CT models.
The explosion of activity in the SERS field started in the late 1970s, whilst the research accelerated in the early 1980s. Unfortunately it turned out that only a few metals, mainly roughened Ag, Au and Cu, provide the greatest enhancement of the Raman effect. These severely limited the breadth of practical applications involving other metallic materials. Moreover, the atomically flat surfaces, commonly used in fundamental research in surface science, are not accessible for SERS investigations. In consequence, the activity in the field has declined since the mid-1980s, and surface Raman spectroscopy is not normally considered as a general analytical tool unlike surface IR spectroscopy which is used widely in surface science, analytical chemistry and materials science.
The situation has changed dramatically with several important progressive steps both experimental and theoretical made in the late 1990s (vide infra). These recent advances lead me to believe that the renewal of extensive SERS study is rapidly approaching, and that the wide spread application in advanced materials, surface processing, catalysis, electrochemistry, corrosion, sensors, biology and medicine will boom. Although IJVS has published papers on SERS, we have never comprehensively explained the process, its origin, experimental details etc. Therefore, Patrick and I both think it could be an appropriate time to edit a special issue on this exciting area, and hopefully to shed new light on this field for the new millennium.
The recent progress (or breakthrough) in the SERS field can be summarized in three parts: SERS substrates, techniques and theories as shown in Figure 1. Their synergetic efforts have advanced research to a high level. Accordingly, this special issue on SERS consists of eight articles reporting or overviewing the recent progress. These articles can be classified into four parts respectively, but with certain degree of overlap: the substrate, technique, theory and applications. The articles are of graded sophistication, aimed at a variety of readers from the non-specialist to the dedicated and experienced spectroscopist. For instance, most of the articles emphasize the experimental details in order to assist readers to prepare specific substrates and perform SERS measurements.
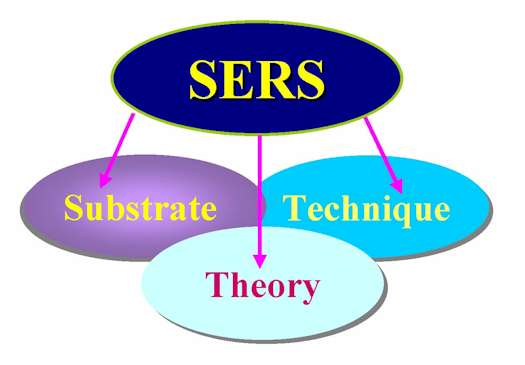
Figure 1.
SERS Substrates
Because SERS activity critically depends on the nature of metal and surface roughness, substrate preparation is always of central concern [7,9,12,17], and hence is the major component in this issue. The two most widely used SERS active substrates are metal colloids (especially aggregated colloids) generated by chemical methods [18] and electrode surfaces roughened by one or more electrochemical oxidation-reduction cycles (ORC) [19]. The other less common substrates include surfaces roughened by chemical etching in acids, “island films” deposited on glass surfaces at elevated temperatures or surfaces finished by electrochemical deposition from solutions, and films deposited by evaporation or sputtering in vacuum onto cold (100 K) substrates. Metal colloids can also be prepared by exposing metal foils to the focused radiation of high power pulse laser in an organic solvent or pure water.
Substrates for Single Molecule Detection
The most significant progress made recently in SERS substrates could be the following observation: the preparation of silver colloids deliberately designed to produce particles of certain size and shape can generate nanoparticles generating with enormous enhancements up to 1014, making single molecule Raman spectroscopy a reality [20-23]. Single molecule imaging and spectroscopy is of basic scientific and practical interest and has been the subject of considerable effort over the past few years. Because there are no articles related to this important subject in this issue, it is necessary for me to give a very brief overview on this aspect.
Recently two Raman groups have achieved this goal recently using very different approaches. Kneipp et al. have reported the detection of single molecules of crystal violet adsorbed on aggregated clusters of colloidal silver [20]. They used near-infrared excitation which is not resonant with any intramolecular optical transitions of the dye but efficiently excites the plasmons of the fractal aggregated silver colloids. Nie and Emory, on the other hand, combined surface and resonance enhancement (SERRS) to produce the required sensitivity to detect a dye molecule adsorbed on the surface of a single silver particle [21].
Nie and Emory used standard citrate reduction techniques to produce silver sols composed of a heterogeneous collection of mostly unaggregated particles of various sizes and shapes. Surprisingly, a small number of these particles showed extraordinarily high enhancements; they have been labeled ‘hot’ particles. These particles can be
identified using a two-dimensional Raman imaging technique and observing the very strong Raman signal of the adsorbed Rhodamine 6G molecule from only certain particles [21]. They have also reported that the relatively intense scattering was observed when the laser polarization was aligned along the long axis of an ellipsoidal particle which is in fact the most polarizable direction. This is in contrast to what has been commonly recognized that SERS is depolarized. Furthermore, in a series of Raman spectra taken from a single molecule at single particles at different times, fluctuations both in frequency and intensity were observed [21].
The results briefly described above clearly illustrate the high potential of SERS among single molecule detection methods, not only for its tremendously high sensitivity, but also its capability of characterizing the molecular structure, orientation and dynamics. For readers interested in a comprehensive review on single molecule Raman spectroscopy, the excellent review by Kneipp et al is strongly recommended [23].
It is of particular interest that only silver nanoparticles with certain size and shapes are optimal for getting extremely high SERS effects, essential for single molecule detection. Combined optical and AFM measurements showed that most of the useful particles were isolated and of dimension ca. 80-100 nm and have shapes which ranged from spherical to rod-like. This evidence clearly implies that ordered nano-structures with narrow size-distributions compose a very promising class of highly active SERS substrates.
This motivation has driven people to fabricate various forms of surfaces and nanostructures. They include ordered nanoparticles hosted by self-assemble layers [24] and nano-wire and nano-rod arrays prepared by template synthesis techniques [25]. The latter are fabricated by electrochemically depositing metals into the nano-holes of the porous alumina film, as shown by Ren et al. It is of special interest that SERS intensities depend critically on the length of the nano-rods emerging from the surface, thereby optimizing remarkably the surface enhancement [18]. The latter are fabricated by electrochemically depositing metals into the nano-holes of a porous alumina film followed by partially dissolving the alumina, as shown in Figure 2. It is of special interest that SERS intensities depend critically on the length of these nano-rods emerging from the surface, thereby optimizing the surface enhancement, as discussed by Ren et al[18].)
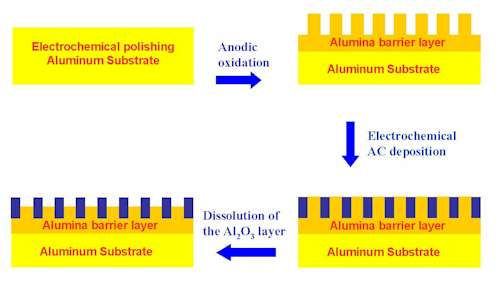
Figure 2. The schematic diagram of the fabrication processes of metal nanorod arrays.
Extending SERS study to a wider range of metallic and non-metallic materials
The strategy of extending the SERS substrate from the coinage metals of Au, Ag and Cu to a wide variety of metallic and non-metallic materials is crucially important for developing SERS as a versatile and powerful tool in surface science and materials science [17, 25]. Therefore, three groups active in this direction have been invited to present overviews and they have contributed the latest development in this issue [18, 26, 27].
Ren et al have comprehensively introduced the background of this topic and then described systematically the experimental detail preparing transition metal substrates and calculating the surface enhancement factor, as well as some practical applications in electrocatalysis and corrosion. The unique ORC roughening procedures (some of them are combined with chemical etching ) have been found particularly essential to create sufficient SERS activity from many important transition metals including Ni, Co, Fe, Pt, Pd, Rh and Ru. The surface enhancement factor ranges from one to four orders of magnitude for transition metals and, depends critically on the nature and surface morphology of the metal. It is now feasible to apply SERS in many fields of practical importance, including electrocatalysis and corrosion [18].
Based on the tactic of “borrowing the SERS effect”, two approaches, have been made to successfully extend SERS studies to a wide variety of materials, not only transition metals but also semiconductors, polymer and organic films, and liquid crystals, that normally show weak- or no SERS activity. The first procedure is commonly named the ‘overlayer’ or ‘thin film technique’, by which SERS active Ag or Au substrates are coated with ultrathin films of other metals or semiconductors. With the aid of the long-range effect of the electromagnetic enhancement created by the SERS active substrate underneath, SERS spectra of adsorbates can be obtained on these films, see Fig. 3a. This approach can also be extended to the study of solid-solid interfaces, through which metal oxides, semiconductors, polymer or organic films are coated in different ways onto the SERS active substrates (Fig. 3b). Strong SERS signals provide meaningful information from solid-solid interfacial structures that are difficult to obtain by other means. For recent progress in the overlayer technique, readers should refer to the excellent review article by Weaver et al. [17].
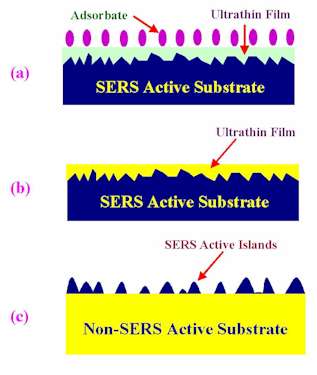
Figure 3. Schemes of various specially designed SERS substrates to study: (a) adsorbate on thin film of non-SERS activity, (b) the thin film/metal interface, and (c) the non-SERS active substrate surface.
The second approach is to reverse the geometry of the overlayer technique, see Fig. 3c. SERS-active metal islands are deposited electrochemically or chemically onto non-SERS active substrates including liquid crystals, metal oxides, semiconductors or polymers, to investigate the surface structures and properties of the materials attaching to and surrounding the SERS-active islands.
Quagliano demonstrates that adsorbed species on semiconductor surfaces can be detected by SERS successfully by using two SERS-activation procedures [26]. One consists of depositing an Ag island film on the semiconductor surface. The other is an especially encouraging one, which is based on the use of a chemical etching process to activate the surface. It has shown for the first time that semiconductors themselves may be capable of supporting the SERS effect. Therefore, it is highly desirable to calculate the surface enhancement factor of the semiconductor in order to confirm the SERS phenomenon quantitatively, thereby revealing potentially the SERS mechanism operated on semiconductors.
As a result of the systematic work in particular the elegant sampling technique, Xue and Lu have demonstrated that SERS can be powerful in the characterization of the polymer/metal interface and the polymer film surface [27]. It almost exclusively enhances the first monolayer of molecules adjacent to the SERS active surface or particle. Measurements at the interface are performed on nitric acid-etched Ag or Cu substrate. When studying the polymer surface Xue and Lu chemically deposited Ag colloids on the surface. SERS measurements on nanometer diameter fibre and liquid crystal films were also performed successfully without disturbing the surface morphology.
The SERS Techniques
It is necessary to point out that most the successful new approaches on SERS substrates mentioned above have been aided by new techniques such as confocal Raman microscopy. In recent years the sensitivity level of surface Raman spectroscopy has been improved substantially with the advent of the confocal microscope, the holographic notch filter and the CCD detector [25]. The confocal Raman microscope provides excellent three-dimensional spatial focusing. It increases the surface scattered light collection efficiency and minimizes the interference from the solution phase. A detailed discussion on the advantages of confocal Raman microscopy is given in the paper of Ren et al [18].
On the other hand, the successful realization of the new SERS technique also requires some assistance from substrate preparation. This is well illustrated by Viets and Hill in their paper on fibre-optic SERS technique [28]. Fibre-optics can avoid hazardous, freely propagating laser light, and permits remote measurements and adjustment-free sample exchange. A combination of fibre-optic probes and SERS, offers both high sensitivity and specifity in analyte molecules as powerful chemical sensors with low cross-sensitivities. When one uses a single fibre for guiding both excitation laser and Raman scattered light, the preparation of properly roughened metal films at the tip of the fibres is required. Several ways have been developed to generate SERS-active fibre tips, including slow evaporation of metals forming island films, vacuum deposition of metal films over nanoparticles, and evaporation of metals on roughened fibre tips. These procedures make the Fibre-optic SERS technique more promising than ever in a wide range of applications [28].
From the fundamental point of view, one of the most important recent developments in the SERS technique is the application of the attenuated total reflection (ATR) method. This method permits the study of very weak signals from single crystal surfaces. Although there have been some reports on surface Raman studies on atomically flat single crystal surfaces, the early studies were restricted to vacuum /solid and air/solid interfaces. Recently, SERS studies on true smooth single crystal electrode surfaces have been accomplished [29]. Futamata describes the method both in principal in practice through introducing his experimental setup and latest results [30].
The detailed comparison of the signal from ordered monocrystalline surfaces and from rough surfaces will be significant because it should achieve a better level of understanding of SERS mechanisms. Moreover, this approach is also very helpful in linking the structural properties of in-situ electrochemical, catalysis and metal-UHV interfaces and aiding and enriching the structural interpretation of Raman data about structurally well-defined surfaces. However, it should be noted that, at present, there is only a very limited number of molecules with large Raman cross section and hence accessible to study. The major challenge to the further application of ATR-Raman spectroscopy is to improve the sensitivity thus enable us to study a range of molecules [30].
Futamata has also introduced a very interesting hyphenated technique – ATR-Raman and scanning near-field optical microscopy (SNOM). This method has a spatial resolution well beyond the diffraction limit (~20 nm). It should provide a powerful tool to obtain chemical information of surfaces (interfaces) in the nanometric scale [30,31]. However, it should be noted that the technique is immature at present and the sensitivity is still very low compared with the conventional Raman system due to extremely low throughput of Raman photons. However, sophisticated further development of the instrumentation is expected to broaden the application of this method.
Combined strategy techniques are particularly helpful in obtaining deeper insight into complicated molecular systems such as Langmuir-Blodgett (LB) monolayers of a new dye molecule. Kam and Aroca have applied several techniques in a systematic study on the LB films fabricated on dielectric and metal substrates [32]. They used transmission and reflection-absorption infrared spectroscopy to determine the long range molecular organisation within the film. SERS and Surface-enhanced infrared (SEIR) were used to identify in detail the interaction of the film with the metal substrate. Moreover, ab initio quantum chemical computations on infrared and Raman intensities and wavenumbers were performed to aid the assignment of the fundamentals. Through their comprehensive work the reader should get an excellent impression of the methodology of the combined techniques [32].
SERS Applications
As has been described above SERS is among the most sensitive techniques available to surface science. Its capability of delivering specific chemical identification and to couple this with a wide range of instruments, has led to its continuing use in both new and traditional areas of surface science. These are of prime importance to both fundamental research and to a range of industrial fields related to surface oxidation, adhesion, corrosion and catalytic processes and in advanced materials, biology and sensor research. Although it is far beyond scope of this issue to cover all these application fields, several papers in this issue do briefly introduce SERS applications.
For those who are not familiar with SERS, it is essential to have a general feel about how to apply SERS for a specific purpose. The paper by Chowdhury et al. is recommended. They used a classical SERS system — silver colloids and provided rich information in their paper through the systematic SERS study on three interesting samples [19]. After describing the ir experimental details, they clearly demonstrate how to apply SERS to reveal in detail surface adsorption of organic molecules at colloids. Changing the solution pH and molecular concentration, one can analyze the relevant SERS spectra so as to identify protonated, depronated and hydrated states, and the orientation of the adsorbate as well as surface adsorption sites and surface coverage [19].
SERS Mechanisms
Several years ago few were fully convinced by statements that SERS enhancement could be as high as 15 orders of magnitude, and SERS active materials would include a variety of transition metals and probably semiconductors. This issue and some recent papers clearly demonstrate that these statements are reality. These observations really challenge our conventional understanding of SERS and provide some key evidence which should establish new theories or reinterpretation as appropriate [22,33].
Because the enhancement mechanism is the most difficult object in the SERS field, the review article by Wu et al is arranged as the last part of this issue [34]. To help readers to comprehend the origin of SERS they start by reviewing the electromagnetic enhanced mechanism in sequence from a flat interface to a small spherical surface, then finally to the fractal colloidal cluster that is the latest advance in the EM theory. The chemical enhanced mechanism and the non-EM mechanism, and the charge transfer (CT) theories are all presented. The most interesting part of this article could be the discussion on the most recent advances in SERS mechanisms. It involves the idea of combinations of the EM and CT theories in order to enable us to explain the huge enhancement up to 15 orders of magnitude. Finally, to assist those who are not familiar with surface vibrational spectroscopy, the surface selection rule for SERS are introduced and discussed, very important to correctly explain the SERS process and to analyze the orientation of surface species [34].
SERS and Nano-science
In the final part of this paper, it is necessary to point out that SERS is an important issue not only for surface science but also for nano-science. At the present stage we realize much more clearly than twenty years ago that SERS is one of the important phenomena of nano-science. Obviously, the interaction of SERS with surface science and nano-science will be one of the central issues in the near future, see Fig. 4. In fact the renewal of great interest and extensive attention to SERS has been stimulated by the rapid development of nano-science in recent years.
Figure 4.
With aid of nano-science (-technology), numerous methods have been developed to syntheses nanoparticles of controlled size and shape and to fabricate nanorod or nanodot arrays, and hence to obtain the SERS active surfaces. The analysis of the SERS spectra from these well ordered nanostructures are much more simple randomly roughened surfaces. These analyses enable us to test the SERS models. In reverse, information derived from SERS can reveal unique optical and electronic properties of nanodots, nanorods and nanowires [35].
Scanning probe microscopy (SPM), is one of most powerful techniques in nano-science, for probing a wide variety of surfaces (interfaces) with atomic- to nano-scales. It is highly desirable to couple SERS and SPM for the optical and morphological characterization [36]. There is no doubt that much useful and reliable information can be obtained if one can combine the advantages of these two techniques to simultaneously study SERS active surfaces for correlating correctly SERS activity and surface morphology. Several studies have been directed along this avenue, including setting up a hyphenated Raman-STM system [36]. They enable us to characterize in detail the nanoparticles and their aggregates.
Moreover, increasingly powerful computers and lasers, such as tunable lasers with excitation lines from the UV to the near IR and pulse lasers with time-resolution from ns to fs, will greatly accelerate developments in SERS theory and experiment. Therefore, it is reasonable to be optimistic that the SERS mechanism can be described quantitatively as the general conclusion in the near future and SERS will become a widely used and powerful technique in surface science and nano-science.
Acknowledgements
The author acknowledges the support of the National Natural Science Foundation of China and the Ministry of Education of China.
Abstract
As the preface of this special issue, this paper aims to provide a general introduction on surface-enhanced Raman spectroscopy (SERS) for wide variety of readers. The history and basic experimental facts are first reviewed concisely. Then very recent fundamental development, including the SERS substrate, technique and theory, and applications are overviewed through introducing eight papers in this issue. Finally the prospective progress is discussed briefly with emphasize on the correlation with nano-science.
References
- M. Fleischmann, P. J. Hendra and A. J. McQuillan, Chem. Phys. Lett., 26, 163 (1974).
- D. J. Jeanmaire and R. P. Van Duyne, J. Electroanal. Chem., 84, 1 (1977).
- M. G. Albrecht and J. A. Creighton, J. Am. Chem. Soc., 99, 5212 (1977).
- R. K. Chang and T. E. Furtak, Surface Enhanced Raman Scattering, Plenum Press, New York (1982).
- R. P. Van Duyne, Chemical and Biochemical Applications of Lasers, edited by C. B. Moore, Vol. 4, p. 101, Academic Press, New York (1979).
- A. Otto, Light Scattering in Solids IV, edited by M. Cardona and G. Gunthrodt, p. 289, Spring-Verlin, Berlin (1984).
- R. K. Chang and B. Laube, Crit, R. C. Rev. Solid State Mater. Sci. 12, 1 (1984).
- M. Moskovits, Rev. Mod. Phys. 57, 783 (1985).
- M. Fleischmann, Proc. Robert A. Welch Found. Conf. Chem. Res., 90 (1986).
- J. A. Creighton, Spectroscopy of Surfaces, edited by R. J. H. Clark and R. E. Hester, p. 37, J. Wiley & Sons, Chichester (1988).
- R. L. Birke and J. R. Lombardi, Spectroelectrochemistry: Theory and Practice, edited by R. J. Gale, p. 263, Plenum Press, New York (1988).
- R. L. Garrel, Anal. Chem. 61, 401A (1989).
- J. E. Pemberton, In-situ Studies on Electrochemical Interfaces: A Prospectus, edited by H. D. Abruna, p. 328, VCH Verlag Chemie, Berlin (1991).
- B. Pettinger, in J. Lipkowski, P.N. Ross (Eds.), “Adsorption of Molecules at Metal Electrodes“, VCH, New York, 285-345 (1992).
- A. Otto, I. Mrozek, H. Grabhorn, W. Akemann, J. Phys.: Condensed Matter, 4, 1143 (1992).
- A. Campion, P. Kambhampati, Chem. Soc. Rev., 27, 241-249 (1998).
- M. J. Weaver, S. Zou, H. Y. H. Chan, Anal. Chem., 72, 38A (2000)
- B. Ren, J.L. Yao, C.X. She, Q.J. Huang, Z.Q. Tian Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 4 (2000).
- J. Chowdhury, M. Ghosh, K. M. Mukherjee and T. N. Misra, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 7 (2000).
- S. Nie, S. R. Emory, Science, 275, 1102 (1997).
- K. Kneipp, Y. Wang, H. Kneipp, LT Perelman, I. Itzkan, R.R. Dasari, M.S. Feld, Phys. Rev. Lett., 78(9), 1667 (1997).
- A.M. Michaels, M. Nirmal, L.E. Brus, J. Am. Chem. Soc., 121, 9932 (1999)
- K. Kneipp, H. Kneipp, I. Itzkan, R. Ramachandra, R.R. Dasari, M.S. Feld, Chem. Rev. 99, 2957 (1999).
- K.C. Grabar, R.G. Freeman, M.B. Hommer and M.J. Natan, Anal. Chem., 67, 735 (1995).
- Z. Q. Tian, J. S. Gao, X. Q. Li, B. Ren, Q. J. Huang, W. B. Cai, F. M. Liu and B. W. Mao, J. Raman Spectrosc., 29, 703 (1998).
- L.G. Quagliano, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 3 (2000).
- G. Xue and Y. Lu, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 5 (2000).
- C. Viets and W. Hill, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 8 (2000).
- A. Bruckbauer and A. Otto, J. Raman Spectrosc., 29, 703 (1998).
- M. Futamata, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 9 (2000).
- V. A. Markel, V. M. Shalaev, P. Zhang, W. Huynh, L. Tay, T. L. Haslett, M. Moskovits, Phys. Rev. B 16, 8080 (1999)
- A. Kam and R. Aroca, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 6 (2000).
- S. Grésillon, L. Aigouy, A. C. Boccara, J. C. Rivoal, X. Quelin, C. Desmarest, P. Gadenne, V. A. Shubin, A. K. Sarychev and V. M. Shalaev, Phys. Rev. Lett. 82, 4520 (1999).
- D.Y. Wu, B. Ren and Z.Q. Tian, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 10 (2000).
- J.L. Yao, G.P. Pan, K.H. Xue, D.Y. Wu, B. Ren, D.M. Sun, J. Tang, X. Xun and Z.Q. Tian, Pure Appl. Chem., 72(1), 221 (2000).
- Z. Q. Tian and B. Ren, Chinese J. Chem., 379, 271(1994).
REF: Z. Q. Tian, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 2 (2000)