Fourier Transform infrared spectroscopic features of human breast benign and malignant tissues
CONTRIBUTED ARTICLE
4. Fourier Transform Infrared Spectroscopic features of human breast benign and malignant tissues
Tiyu Gao, Yunxiang Ci
Department of Chemistry
Peking University,
Beijing, 100871,
People’s Republic of China
Tel: 0086-010-62757908
Fax: 0086-10-62751708
Email:gaoty [at] chemms.chem.pku.edu.cn
ciyx [at] chemms.chem.pku.edu.cn
Abstract
Some remarkable spectral differences are observed among the normal, hyperplasia, fibroadenoma and invasive infiltrating ductal carcinoma breast tissue samples:
(i) characteristic spectral patterns of fibroadenoma, and carcinoma tissues appear in the frequency regions of 950~1150 cm-1 and 2800~3050 cm-1,
(ii) peak near 970 cm-1 is sharper and stronger, the series of bands near 1204cm-1, 1280cm-1 and 1338cm-1 are weaker and broader for carcinoma tissue whereas the band near 970 cm-1 is weaker than that for benign tissues,
(iii) the band near 1163 cm-1 band in benign tissues shifts to 1171 cm-1 in carcinoma tissue,
(iv) A1032/A1083 and A2958/A2853 ratios in carcinoma tissue are the smallest, whereas are the highest in fibroadenoma tissue among the corresponding ratios, and (v) A1459/A1241 >1.0 for normal tissue, A1453/A1239 >1.0 for fibroadenoma, A1456/A1239<1.0 for hyperplasia and carcinoma tissues. And these spectral differences have important implications not only for evaluating the histological types and grades of breast diseases, but also for probing and analyzing the developing process of the breast lesion at the molecular level.
Keywords
Infrared spectroscopy, breast benign and malignant tissues, collagen
Introduction
Proteins, nucleic acids, lipids and carbohydrates are the important compositional, structural and functional bio-macromolecules in biological cells and tissues. The change of the status of cells, such as the malignant transformation, induces not only the changes of the relative contents of bio-molecules, but also the changes of the structures and conformations of bio-molecules due to the perturbation of the intermolecular and intramolecular interactions. Fourier transform infrared (FTIR) spectroscopy, which is used to measure the vibrational modes of the functional groups of molecules, is sensitive to molecular structure, conformation and environment. It is possible to use the sensitivity of FTIR spectroscopy to detect the biochemical alterations of various forms of neoplastic cells and tissues [1-3]. The purpose of this study is to investigate the compositional and structural differences of benign and malignant breast tissues based on the analysis of FTIR spectral differences. We report our findings and interpret our results in terms of spectral profiles, absorption frequencies and the relative intensity. [4]
Experimental and Methods
Samples and procedures
Human breast tissues were obtained from surgical specimens at People’s Hospital, Beijing Medical University. Each sample was cut into two pieces, the first was sent to the pathologists for evaluation through standard pathological methods; the second was washed with 0.9% NaCl solution and frozen in liquid nitrogen immediately after collection until used for IR spectroscopic studies. Small amounts of the frozen thin tissue sample was evenly spread over the surface of a specially designed infrared window of BaF2, then the sample was placed in a vacuum desiccator and allowed to dry to form a even film. Among 54 samples, 14 samples were diagnosed cytologically as normal, 10 samples were as hyperplasia, 13 samples as fibroadenoma, 16 samples as invasive infiltrating ductal carcinoma.
Apparatus and data analysis
Infrared spectra in 400-4000 cm-1 region were recorded on a Bruker Vector 22 Fourier transform spectrometer equipped with an air-cooled DTGS detector. Infrared window of BaF2 without cell suspension was scanned as the background, for each spectrum, 512 scans were co-added at a spectral resolution of 2 cm-1 . To minimize problems from avoidable baseline shifts, the spectra were baseline-corrected. After picking the peaks, the values of the absorbance of the corresponding peaks are obtained.
Results
Assignment of infrared absorption bands human breast tissues
The representative infrared spectra of the tissues in the frequency regions of 950 to 1700 cm-1 and 2800 to 3050 cm-1 are shown in Figure 1 and Figure 2 respectively. The assignment of infrared bands is achieved with the combination of the previously well-established IR spectra of cellular organelles and bio-molecules isolated from tissues and cells [1, 5-7] and the understanding of the composition and properties of tissues. Benign fibrocystic disease involves ductal epithelia proliferating cystic changes and a dominant collagenous component in the stroma. For example, fibroadenoma is composed of both an epithelial component and a substantial stromal neoplastic element with collagen deposition. The ductal carcinoma is associated with stromal fibroblastic proliferation and collagen deposition in one way, related to the increase of DNA, RNA from the rapid synthesis of nucleic acids and the glycolysis of carbohydrates in another way. Breast tissue is mainly consisted of epithelial components and the connective tissue whose dominant protein is collagen. The spectrum of breast tissue can be considered to be mainly from the overlapping of the epithelial cells and collagen in the connective tissue.
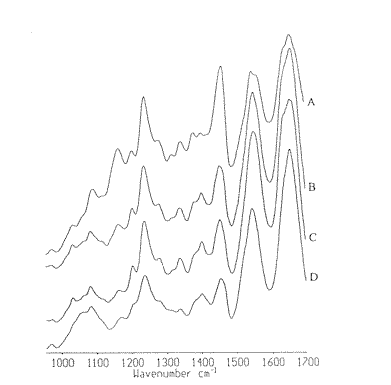
Figure 1. Infrared spectra in the frequency region 950 to 1700cm-1 of human breast tissues:
A : normal; B: hyperplasia C: fibroadenoma and D: invasive ductal carcinoma
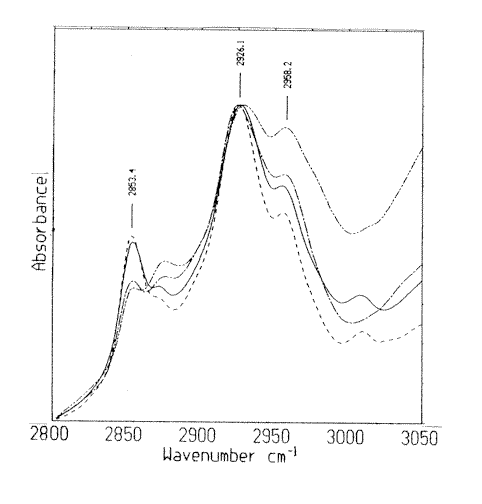
Figure 2. Infrared spectra in the frequency region 2800 to 3050cm-1 of human breast tissues.
A: normal; B: hyperplasia; C: fibroadenoma and D: invasive ductal carcinoma
As shown in Figure 1 and Figure 2, the weak and broad peak near 970 cm-1 is generally assigned to the symmetric stretching mode of dianionic phosphate monoester of cellular nucleic acids 8,9] . The bands near 1083 cm-1 and 1240 cm-1 are generally assigned to the symmetric and asymmetric phosphate (PO2–) stretching modes (us PO2– and uas PO2–) respectively. The peaks at 1204cm-1, 1280 cm-1 and 1338 cm-1 emerge in all spectra of samples, and their relative intensities simultaneously increase or decrease. The simultaneous change indicates that these peaks are originated from the same molecular. In addition, the spectral pattern is significantly similar with that of collagen [10,11]. The vibrational modes of collagen carbohydrates residues appear at 1032 cm-1 and 1083 cm-1, the amide III/CH2 wagging vibrations of collagen appear at 1204cm-1, 1237 cm-1, 1280cm-1, 1318 cm-1 and 1338 cm-1 [11]. Therefore, the peaks near 1083 cm-1 and 1239 cm-1 can be considered to be from both collagen and nucleic acid. The band near 1163 cm-1 can be assigned to the C-O stretching modes of the C-OH groups of proteins. The peaks at 1400 cm-1 and 1456 cm-1 are mainly arising from the symmetric and asymmetric bending vibrational modes of methyl groups of proteins(ds CH3 and das CH3 ), respectively. Peaks near 2853 cm-1 and 2925 cm-1 are originated from ds CH2 and das CH2, 2959 cm-1 is from das CH3 of proteins, lipids and DNA.
Comparison among the spectra of breast tissues
Comparison of the absorption frequency
From the spectra in Figure1, Figure 2 and Table 1, some striking differences of spectral patterns and parameters can be observed among these tissues. The predominant differences include the following findings. The average absorption frequencies of some absorption bands are listed in Table 1. No obvious differences were found among the frequencies of absorption bands near 970 cm–1 and 1083 cm-1 of these four types of tissues. The infrared spectra of normal and hyperplasia samples in the C-O stretching region consist of a relatively narrow and symmetric band near 1163 cm-1 (Figure 3), whereas the fibroadenoma and carcinoma samples appear as a relatively broad and asymmetric bands whose peaking absorption frequency are near 1163 cm-1 and 1171 cm-1, respectively.
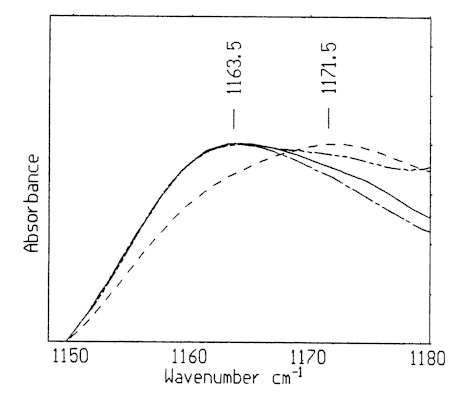
Figure 3. Infrared spectra of breast tissues in the C-OH stretching region of human breast tissues.
A: normal; B: hyperplasia; C: fibroadenoma and D: invasive ductal carcinoma
Compared to the benign tissue samples whose absorption frequencies are almost in the same region, the shift of about 8cm-1 of the frequency in the carcinoma sample was observed, and the shift indicates the increase of malignant epithelial tissue in carcinoma tissue, because the band near 1163 cm-1 is normally assigned to the stretching mode of C-O groups of proteins in normal connective tissues, the band near 1171cm-1 band is mainly from malignant epithelial cells.[10] In addition, the average frequencies of absorption band near 1239 cm-1 of the abnormal tissues are almost in the same region (Table 1).
| frequency(cm-1) | cases | 970 | 1083 | 1163 | 1240 |
| normal | 14 | 970.1+ 0.3 | 1083.5+ 0.3 | 1163.8+ 0.5 | 1241.2+ 0.3 |
| hyperplasia | 10 | 971.1+ 0.6 | 1083.2+ 0.6 | 1162.3+ 0.5 | 1238.3+ 0.2 |
| fibroadenoma | 13 | 971.0+ 0.4 | 1081.9+ 0.6 | 1162.4+ 0.3 | 1238.5+ 0.1 |
| carcinoma | 17 | 970.2+ 0.7 | 1083.5+ 0.4 | 1171.3+ 0.7 | 1238.9+ 0.5 |
Table 1. Average frequencies(mean+SD) of the major absorption bands of breast tissues
Comparison of the absorbance ratios of the major absorption bands
The relative intensity of major absorption bands are also important spectral parameters in obtaining the semi-quantitative information about the relative contents of bio-molecules among these tissues. The ratios of the absorbance of the bands concerned were calculated and listed in Table 2. As shown in Figure 1, the band at 970 cm-1 in carcinoma spectrum is sharper and stronger than that in non-carcinoma spectra. This result implies the increase of the relative content of nucleic acids in carcinoma tissue. The peaks at 1204 cm-1, 1280 cm-1 and 1338 cm-1 are weaker and almost disappears in carcinoma tissue than that in non-carcinoma tissues. The result indicates that the relative levels of collagen in carcinoma tissue is lower than that in hyperplasia and fibroadenoma tissues. These results are consistent with that of the histopathological examination. Thirdly, among the average absorbance ratios of A1032 / A1083 (Table 2), the value of such ratio of carcinoma tissue is the smallest and localized in a separate range compared with non-carcinoma tissues while fibroadenoma tissue has the highest ratios. This result implies the increase of the relative content of nucleic acids in carcinoma tissue and the slight increase of collagen in fibroadenoma tissue. Fourthly, the ratios of A1457/A1239 corresponding to the normal and fibroadenoma tissues are localized in the ranges of 1.08-1.20 and 1.01-1.17, respectively, closer and substantially higher than the range of 0.62-1.04 for hyperplasia and carcinoma tissues. This analysis may indicate the slight increase of nucleic acids in carcinoma tissue. Finally, the ratios of A2958/A2853 of normal and hyperplasia tissues are in the same region, whereas the ratio in fibroadenoma is the highest and shows a wide range of variations. In the case of carcinoma tissue, A2958/A2853 ratio is the smallest and closest. This trend might indicate an increase in number of methyl groups compared to methlene groups in fibroadenoma tissue, which is in agreement with the increase of protein fibers.[12] The decrease of A2958/A2853 ratio in carcinoma tissue may result from the increase of malignant epithelial components because the A2958/A2853 ratio of malignant epithelial cells is the smallest among normal connective tissue, normal and malignant epithelial tissues.[11]
| cases | A1032/A1083 | A1457/A1239 | A2958/A2853 | |
| normal | 14 | 0.70+ 0.12 | 1.12+ 0.04 | 1.24+ 0.12 |
| hyperplasia | 10 | 0.75+ 0.09 | 0.84+ 0.19 | 1.28+ 0.15 |
| fibroadenoma | 13 | 0.83+ 0.03 | 1.09+ 0.08 | 1.87+ 0.30 |
| carcinoma | 17 | 0.44+ 0.10 | 0.83+ 0.19 | 1.07+ 0.06 |
Table 2. Average absorbance ratios (mean+SD) of the major absorption bands of breast tissues
Discussion
The results above show that normal, hyperplasia, fibroadenoma and invasive ductal carcinoma tissues have their own characteristic spectra and spectral parameters. Some remarkable differences exist between the spectra of these four types of breast tissues in terms of spectral profiles, absorption frequencies and the absorbance ratios of the major absorption bands. In carcinoma tissue, a higher level of nucleic acid and lower level of collagen were observed. However, in fibroadenoma tissue, a higher level of collagen was observed. These results, which are in accordance with histopathological observation, such as the nucleus condensation in carcinoma tissue and the collagen deposition in fibroadenoma tissue, may imply that the states of breast disease depend on, in some extent, the alteration of composition of the connective tissues. These methods of analysis above result in highly corrections between spectra and the four types of breast tissues. Whether it is a powerful tool for differentiation of all types of breast tissues, detailed analyses of a number of tissues types will be required.
Acknowledgements
This project was supported by National Natural Science Foundation of China.No. 39730160.
References
- E. Benedetti, L. Teodori, M. L. Trinca, P. Vergamini, F. Salvati, F.Mauro, and G. Spremolla, Appl. Spectrosc., 1990, 44, 1276.
- B. Rigas, P. T. T. Wong. Cancer Research, 1992, 52, 84.
- T J O’Leary, W F Engler, K M Ventre. Appl. Spectrosc., 1989, 43, 1095.
- Y. X. Ci, T. Y. Gao, J. Feng, Z. Q. Guo, Appl. Spectrosc., 1999, 53, 512.
- P. T. T. Wong, “High Pressure Vibrational Spectrocopic Studies of Aqueous Biological System: from Model System to Intact Tissue” in R. Winter and J. Jonas, Eds. “High Pressure Chemistry, Biochemistry and Material Science“, Kluwer Acedemic Publishers, Dordrecht, 1993, p.511.
- P. T. T. Wong, S. M. Goldstein, R. C. Greckin, T. A. Godwin, C. Pivik, and B. Rigas, Cancer Research, 1992, 53, 762.
- F. S. Parker, “Application of Infrared Spectrocopy in Biochemistry, Biology and Medicine“,Plenum Press, New York, 1971.
- M. F. K. Fung, M. K. Senterman, N. Z. Mikhael, S. Lacelle, P. T. T. Wong, Biospectroscopy, 1995, 2, 155.
- H. Susi, “Infrared Spectra of Biological Macromolecules and Related Systems“,in S. N. Timasheff and C. D. Fasman, Eds. “Structure and Stability of Biological Macromolecules“, Marcel Dekker, New York, 1969, p.641.
- P. T. T. Wong, S. Lacelle, H. M. Yazdi, Appl. Spectrosc., 1993, 47, 1830.
- M. Jackson, L. P. Choo, P. H. Watson, W. C. Halliday, H. H. Mantsch, Biochem. Biophys. Acta, 1995,1270,1
- Z. Q. Tang, “Modern Oncology“, Shanghai Scientific & Technical Publishers, Shanghai, 1993, p.42.
Received 9th July 1999, received in revised format 18th August,
accepted 22nd August 1999
REF: T. Y. Gao, Y. X. Ci Int. J. Vib. Spect., [www.irdg.org/ijvs] 3, 5, 4 (1999)