The identification of pigments in paper coatings by infrared spectroscopy
Sharon D. Wightman and
Alison Murray,
Art Conservation Program,
Department of Art,
Queen’s University,
Kingston,
Ontario, Canada
K7L 3N6
Herbert F. Shurvell
Department of Chemistry,
Queen’s University,
Kingston,
Ontario, Canada
K7L 3N6
Introduction
Coatings on coated papers contain pigments, an adhesive or binder and various additives, which facilitate the coating process. The latter are present in minor amounts. Modern coatings are of two general types: pigmented or functional. For the improvement of printing and graphic reproduction, pigment coatings level the microscopic irregularity of the paper surface and make it more uniformly receptive to printing inks. Functional coatings are applied for various specific purposes. For example, in the food packaging industry, functional coatings provide barriers to fluids and grease, and retard the oxidation of package contents.
Coated papers have been known for hundreds of years. In the fifteenth and sixteenth centuries, artists used coatings of animal glue and calcium carbonate on single sheets of handmade paper. The traditional inventory of pigments was restricted prior to the nineteenth century to calcium carbonate and kaolin (china clay). Other materials, which became available in the twentieth century, include: blanc fixe (precipitated barium sulphate), satin white (calcium sulphate), titanium dioxide, talc, and aluminum trihydrate. In recent years, use of the traditional binders, gelatin and animal glue has declined in favour of casein, latex, starch, soya protein, and natural adhesives. Other materials found in paper coatings include urea-formaldehyde and styrene-butadiene polymers, polyvinyl alcohol and modified celluloses.
A coated paper cannot always be detected visually. Calendared finishing processes give gloss and smoothness to both coated and uncoated papers. Both the presence of and the type of paper coating are important considerations that affect conservation treatment . Various methods for the identification of pigments and binders in coatings are available. These include chemical spot tests for protein starch and cellulose, coating removal and analysis, and ashing for the preparation of samples for detection of inorganic pigments.
Various Infrared spectroscopic techniques have been used for the analysis of paper and paper coatings. Most of the reported infrared studies employed the Attenuated Total Reflection (ATR) technique. Other reported methods include infrared microscopy, and a transmission method in which samples were crushed between diamond anvils. The use of near infrared spectroscopy has also been reported. Using ATR-FT-IR spectroscopy, historical papers have been characterized by their gelatin (hide glue) content and the determination of polymer and mineral content of coatings on papers has been reported. In this paper a simple sampling method is described using infrared transmission measurements for the identification of the main components of coatings on paper.
Samples and Sample Treatment
Table 1 lists the various papers examined in this study. Samples were taken from several general types and weights of papers: bond typing paper, magazine, wallpaper, art stock, gift wrap, post card, and printed catalogue papers. The papers ranged in age from late nineteenth century to the 1990’s. Some of the papers were unused. The typing paper was taken from a pad of sheets. The 1990’s wallpapers were cut from store rolls. The unpasted pre-1970’s wallpaper had been used as a drawer lining. Both of the coloured art stocks were from storage inventories. The remaining papers had been in circulation.
| Sample | Description |
| Typewriter paper erasable bond | Early 1980’s ,white medium weight, 8.5″ x 11″ National Blank Book Co. Inc., Holyoke, Mass. Sample taken from an unused pad. |
| Magazine paper “Country Life” | Vol. CLVII, No. 4053, March 6, 1975, page 547. Published in the U.K. High quality paper. Sample taken from sheet 31.5 cm x 23.5 cm. |
| Magazine paper “Maclean’s” |
December 22, 1997, page 72. Published in Canada.Lower quality paper from a weekly magazine. |
| Wallpaper pre-1970 |
Manufacturer unknown. Unused as wall covering.Floral pattern, sample scraped from ivory coloured background. |
| Wallpaper patterned |
Unused sample of 1990’s plastic coated wallpaper. Supplied by “Color Your World”. Sample scraped from beige-coloured background. |
| Wallpaper coloured |
Unused sample of 1990’s plastic coated wallpaper, with all-over abstract floral print. Supplied by “Color Your World.” |
| Art stock blue card |
Date and manufacturer unknown. Heavy weight glossy surface coated on one side only. |
| Art stock brown paper |
Date and manufacturer unknown. Lightweight coated brown on one side only. |
| Gift wrap paper | 1990’s paper of German origin. Recto printed with floral multi-coloured print with a glossy surface. Verso has a white matte finish, |
| 1897 postcard | Pre-stamped Canada Post issue commemorating the Jubilee of Queen Victoria |
| Furniture catalogue circa1920 | Undated, furniture styles indicate 1920’s. Supplement to catalogue No.54, The Knechtel Furniture Co., Hanover, ON |
| Christmas card circa 1990 |
Glossy coating on one side. Sample taken from glossy side. |
Table 1. Details of papers studied
To obtain a sample for infrared spectroscopy, 1-2 milligrams of material is scraped, with a stainless steel scalpel, from the surface of selected areas of the paper. Printed papers should be scraped in uninked areas such as margins. All papers should be scraped, where possible, on interior surfaces well away from the edges, to reduce the amount of contamination, which might be present from handling and exposure. The scraping must be done lightly and with even pressure in one direction only. The scrapings are transferred to an agate mortar and approximately 200 milligrams of dry KBr powder is added. Scrapings and KBr are ground together until the sample is dispersed homogeneously. The mixture is transferred to a stainless steel die between two 13mm diameter polished anvils, which are wiped free of contamination with lens tissue. Air is evacuated from the die using a vacuum pump. A transparent, or nearly transparent pellet, is obtained, by pressure using a hydraulic press. An article on making good KBr discs can be found in Volume 1/Edition 1 of IJVS [www.irdg.org/ijvs].
In addition to the sample spectra, a set of reference spectra of known coating materials was also recorded. Samples of reference materials were obtained from the paper conservation laboratory at Queen’s University and from various commercial suppliers. The reference spectra were recorded of seven common pigments, five protein binders, four different cellulose samples and four starches.
Infrared Spectra
Infrared transmission spectra of the samples are recorded at a nominal resolution of
4 cm-1 . Baseline fitting, plotting and other manipulation such as overlaying of spectra usually can be carried out using the software available on a modern FT-IR spectrometer. Baseline fitting is needed, because the spectra obtained from KBr discs often have a rising baseline between 4000 and 2000 cm-1. Alternatively the spectra can be transferred to a desktop computer and these manipulations performed using commercial software such as the GRAMSTM software package.
Attenuated Total Reflection (ATR) spectra can be recorded using a KRS-5 crystal with ends cut at 45o. The infrared radiation is directed into the crystal and collected as it leaves by means of mirrors. The spectra reported in this article were obtained using an RIIC Model four-mirror system. The crystal is mounted in a stainless steel holder with strips of the paper sample clamped against each side. A more up to date technique, diamond ATR, is available these days. (See IJVS Volume 2 Edition 2 [www.irdg.org/ijvs])
Reference Spectra
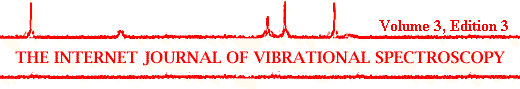
Infrared spectra of seven inorganic materials commonly used as pigments are shown in Figure 1. Infrared spectra of four protein materials used as binders are shown in Figure 2. Figures 3 and 4 show the infrared spectra of three celluloses and four starches.
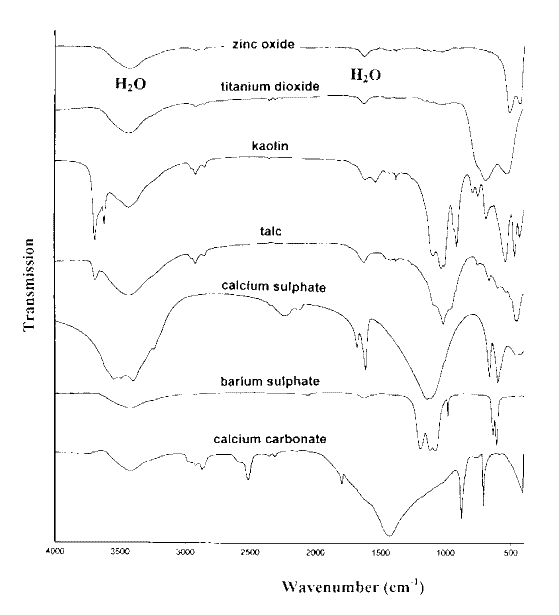
Figure 1.The infrared spectra of seven pigments.
Pigments
Table 2 summarises the characteristic features of the infrared spectra of the seven pigments of Figure 1. All spectra show evidence of the presence of water (a strong broad band at ~3400 cm-1 and a weaker band at 1635 cm-1). It is very difficult to avoid moisture and traces of water are usually present in a sample. Although the KBr is dried in an oven and kept in a dessicator, traces of water are still evident in the infrared spectrum.
| Compound | Wavenumbers (cm-1 ) and description |
| Zinc oxide (ZnO) | 427(s), 510(vs). These are the only peaks due to ZnO |
| Titanium dioxide (TiO2 ) |
528(vs), 690(vs). These bands are stronger and broader than those in the ZnO spectrum |
| Kaolin (china clay) | 431(w), 470(m),540(s), 912(s,sharp), 1030(vs,broad), 3620(m)/3695(s), sharp doublet |
| Talc (talcum, steatite) | 450(s), 1020(vs, broad), 3695(w, sharp) |
| CaSO4 (gypsum, satin white) |
600(s)/665(m), doublet, 1130(vs, broad), 1618(m)/1683(w) sharp doublet |
| BaSO4 (blanc fixe, barite, baryta) | 610(m)/665(s), doublet, 982(w, sharp), 1081/1117 (vs, doublet), 1191(vs) |
| Whiting (CaCO3) | 771(m), 875(s), two very sharp peaks, 1420(vvs), a very broad band, 1800(vw), 2510(w) two sharp peaks |
| Water (H2O) | 1625(w), ~3400(S,broad). These bands are present in all spectra |
Table 2. Characteristic features of infrared spectra of some pigments.
The two oxide pigments ZnO and TiO2 and talc have very simple spectra. Kaolin has a more complex spectrum, with several characteristic peaks. The most notable feature of the infrared spectrum of a coating containing kaolin is a sharp doublet at 3620/3695 cm-1, which is due to stretching vibrations of OH groups in the kaolin structure [10]. Confirmation of the presence kaolin is found from the presence of three peaks of decreasing intensity at 540, 470 and 430 cm-1 and a strong broad band centred near 1030 cm-1.
The two compounds CaSO4 and BaSO4 have broad absorption bands near 1100 cm-1 and several other sharp characteristic peaks in their spectra. CaCO3 occurs in two polymorphic forms, calcite and aragonite. The infrared spectrum of the pigment, whiting has a very strong, very broad band centred near 1420 cm-1. This feature is characteristic of the calcite form of CaCO3.
Binders
Commonly used binders include gelatin, casein, hideglue and soya. These materials are all proteins and as can be seen in Figure 2, their infrared spectra are all very similar. The very strong, very broad absorption between 3700 and 2700 cm-1 and the very strong band centred near 1700 cm-1 are characteristic of proteins.

Figure 2. The infrared spectra of four protein binders.
Celluloses
Celluloses are carbohydrates (polysaccharides) and the main features of their spectra are due to the numerous OH groups in the structure. The very broad, very strong absorption between 3700 and 3200 cm-1 seen in Figure 3 is due to stretching vibrations of these groups and the OH stretching modes of water. The very strong absorption between 1200 and 1000 cm-1 is attributed to stretching of the many C-OH and C-O-C bonds in the structure. Subtle differences in the spectra can be seen between 3000 and 2800 and 1500 and 1300 cm-1. These differences arise from the different arrangements of the methyl and methylene groups in the various celluloses.

Figure 3.The infrared spectra of three celluloses
Starches
Starches are also carbohydrates (polysaccharides) of the general formula (C6H10O5)n and the main features of their spectra are very similar to those of cellulose. Starch occurs in plant cells as structural granules, which consist of concentric shells containing linear amylose, and highly branched amylopectin polymers. The very broad, very strong absorption between 3700 and 3200 cm-1 seen in Figure 4 is due to stretching vibrations of OH groups and the very strong absorption between 1200 and 1000 cm-1 is attributed to stretching of the C-OH and C-O-C bonds in the structure. The infrared spectra of the four starches shown in Figure 4 indicate that the composition of starch is the same regardless of the source.
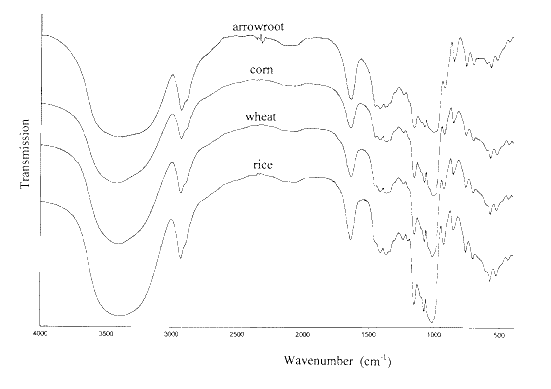
Figure 4.The infrared spectra of four starches
Sample Spectra
Figure 5 shows the infrared spectra of three different papers. A variety of pigment materials are evident in these spectra. Figure 6 shows the spectra of 3 different wallpapers. It is clear from the absence of absorption near 1720 cm-1 that the pre-1970 wallpaper has no plastic coating. Figure 7 compares the spectra of two different coloured card stocks. Figure 8 compares the printed and unprinted sides of a wrapping paper. Figures 9 and 10 show infrared spectra of two old papers. Detailed interpretation of these sample spectra is given below.Sample Spectra
Magazine and erasable typing papers
All three spectra of Figure 5 show peaks due to kaolin. However, there is clearly less of this pigment in the erasable typewriter paper than in the two magazine papers. The strong band at 1420 cm-1 and the sharp peak at 875 cm-1 in the infrared spectrum of the “Country Life” paper show that the coating contains whiting (CaCO3). The sharp peaks near 1720 cm-1 in the spectrum of this paper and that of the typewriter paper indicate that the coatings also contain a synthetic polymer material.
Figure 5. The infrared spectra of the coatings on an erasable typewriter paper and papers from Maclean’s and Country Life magazines.
Wallpapers
Figure 6 compares the infrared spectra of two modern plastic coated wallpapers (“patterned” and “coloured”) with a pre-1970 product. Features of both of kaolin and whiting (CaCO3) pigments are clearly seen in the spectra. The sharp doublet at 3620/3695 cm-1 and the three peaks between 540 and 430 cm-1 identifies kaolin and the strong broad band near 1420 cm-1 and the sharp peak at 875 cm-1 is characteristic of calcium carbonate. There is also evidence for the presence of an organic polymer (peak near 1720 cm-1 ). However, the spectra show that, the coating on the “patterned” wallpaper does not contain kaolin, while the pre-1970 product contains neither whiting, nor polymer.
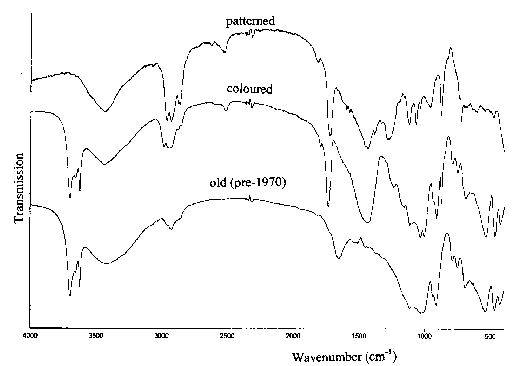
Figure 6. The infrared spectra of the coatings on three wallpapers.
Coloured card stocks
Figure 7 shows that the coatings on these two materials both contain kaolin, (doublet at 3620/3695 cm-1 and three peaks between 540 and 430 cm-1 ), and a synthetic polymer (peak near 1720 cm-1 ). The coating on the “brown” card also contains whiting as indicated by the strong broad band near 1420 cm-1 and the sharp peak at 875 cm-1.

Figure 7. The infrared spectra of the coatings on two card stocks.
Printed wrapping paper
Figure 8 compares the infrared spectra of the printed side and the unprinted side of a wrapping paper of German origin. It is clear from the spectrum that the coating on the printed side contains both kaolin and whiting. In addition, there is evidence for the presence of a polystyrene-containing polymer. This evidence includes three weak peaks in the CH stretching region above 3000 cm-1, the sharp peak at 755 cm-1 and the pattern of very weak absorbances between 2000 and 1700 cm-1 . These features are all present in the infrared spectrum of polystyrene. The spectrum of the unprinted side is essentially that of cellulose with a very small amount of kaolin.
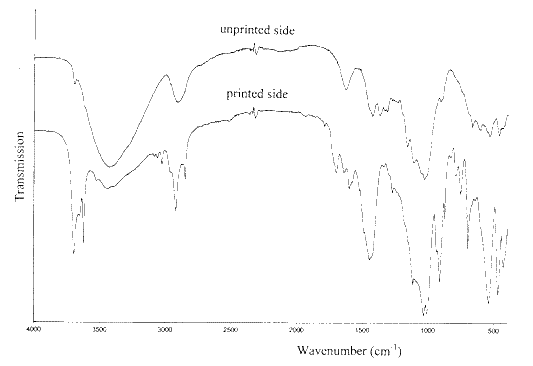
Figure 8. The infrared spectra of both sides of a printed wrapping paper.
A 1897 postcard
Figure 9 shows the infrared spectrum of the coating scraped from an 1897 Canadian postcard (celebrating the Jubilee year of Queen Victoria’s reign). A spectrum of kaolin is included to show that a very small amount of this pigment present. The spectrum of the postcard is essentially that of cellulose.
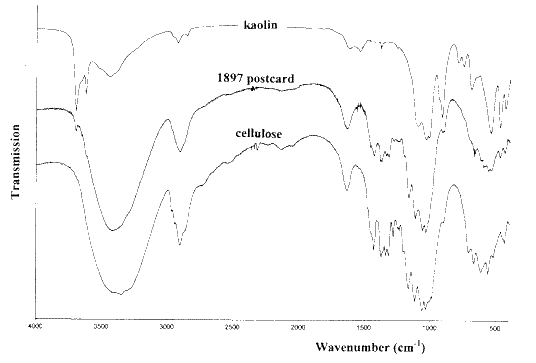
Figure 9. The infrared spectrum of the coating on a 1897 postcard, compared with the spectra of kaolin and cellulose
An old furniture catalogue (circa 1920)
The spectra of Figure 10 show that the coating on the paper taken from a furniture catalogue (circa 1920) contains two pigments BaSO4 and CaCO3. Although CaCO3appears to be present in the coating (the strong broad band near 1420 cm-1 , and the sharp peak at
875 cm-1 ), it is interesting that the calcite peaks at 771 and 1800 cm-1 are absent from the spectrum of the paper coating. This may be due to the form of the “whiting” pigment used at the time.

Figure 10. The infrared spectrum of the coating on the paper of a circa 1920 furniture catalogue compared with the spectra of calcium carbonate and barium sulphate..
Comparison of infrared ATR and transmission spectra
Figures 11 and 12 show the infrared spectra of samples of a coated wallpaper and a Christmas card recorded using both the ATR and the KBr pellet methods. In these Figures baselines have been fitted to the ATR spectra. It is seen that apart from the poorer signal-to-noise in the ATR spectra, the main features of the spectra are similar. The same conclusions concerning the pigments present in the coatings can be drawn from either spectrum. By reference to the infrared spectra of the pigments of Figure 1, it can be seen that both coatings contain kaolin and calcium carbonate. However there are also some obvious differences, which can be attributed to the differences between the transmission and ATR techniques. ATR examines only the top few microns of the surface. On the other hand, the sample for the KBr pellet is manually scraped from the surface and undoubtedly contains some sub-surface material.
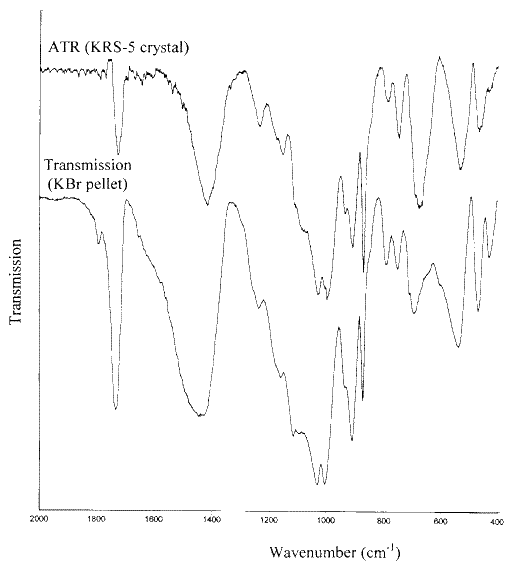
Figure 11. A comparison of the infrared ATR and transmission spectra of the coating on a plastic-coated wallpaper.
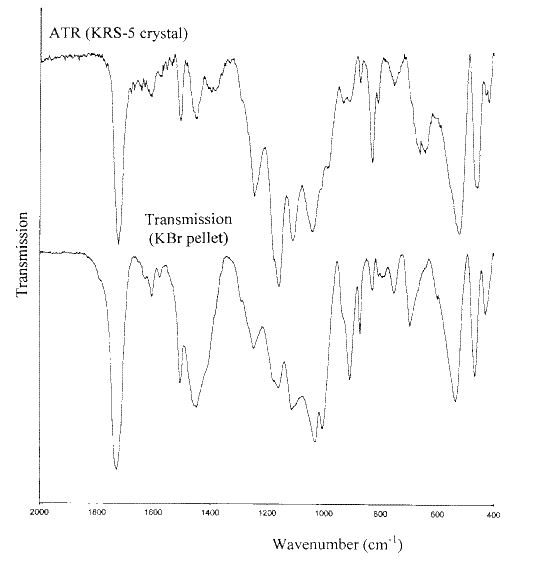
Figure 12. A comparison of the infrared ATR and transmission spectra of the coating on a Christmas card.
Summary & Conclusions
Table 3 summarises the main inorganic pigments found to be present in the coatings of the paper samples studied.
| Sample paper | Pigments identified and comments |
| Typewriter paper erasable bond | polymer, kaolin (minor component) |
| Magazine paper “Country Life” | kaolin, whiting, polymer |
| Magazine paper “Maclean’s” |
kaolin only |
| Wallpaper (pre-1970) |
Kaolin only ( similar to Maclean’s) |
| Wallpaper patterned |
whiting, polymer (no kaolin) |
| Wallpaper coloured |
kaolin, whiting, polymer |
| Art stock blue card |
kaolin, polymer |
| Art stock brown paper |
kaolin, whiting, polymer |
| Gift wrap paper | kaolin, whiting, polystyrene polymer unprinted side: cellulose, kaolin |
| 1897 postcard | cellulose, with small amount of kaolin |
| Furniture catalogue circa 1920 | whiting, baryta (BaSO4), no kaolin |
| Christmas card circa 1990 |
kaolin, whiting, polymer |
Table 3. Pigments identified in coatings on the sample papers
The procedure described here offers a simple rapid method for the qualitative analysis of pigments and other compounds in paper coatings. Information is readily obtained on both organic and inorganic compounds present in the coating materials. Other materials such as synthetic polymers also have been observed in the coatings.
The spectra recorded in this study generally did not reveal the presence of materials such as protein binders and starches. Infrared spectra of these materials are relatively weak and the broad features of their spectra form part of a general background. For older papers, which have little or no pigment coating, the infrared spectrum is essentially that of cellulose.