Application of infrared emission spectroscopy to the study of natural and synthetic inorganic materials
CONTRIBUTED ARTICLE
Ray L. Frost, Huada Ruan and J. Theo Kloprogge
Centre for Instrumental and Developmental Chemistry,
Queensland University of Technology,
2 George Street,
GPO Box 2434, Brisbane,
Q 4001, Australia.
Keywords
dehydration, dehydroxylation, infrared emission, FTIR, FT Raman, phase change, thermal transformation.
Abstract
Infrared emission spectroscopy is a novel technique, which allows the spectra of inorganic materials to be obtained in situ at elevated temperatures. The hot sample becomes the source. High quality spectra with good signal to noise ratio may be obtained using thin films or films of micronised powders. Sets of spectra from 100 to 800 ° C and higher at an appropriate temperature intervals may be obtained. These spectra enable thermal transformation phenomena such as dehydration, dehydroxylation and phase changes together with changes in molecular structure to be studied. Application of the IES technique to naturally occurring minerals, synthetic minerals, and inorganic materials are shown. Advantages and disadvantages of the technique are discussed.
Introduction
Among the vast range of techniques used in vibrational spectroscopy, is the little used infrared emission spectroscopy. This method is well known for its use in scientific studies of stars and planets. However, the technique has not been widely used as a laboratory method in the determination of molecular structure and the changes in the infrared spectrum upon thermal treatment. The measurement of discrete vibrational frequencies emitted by thermally excited molecules is known as Infrared Emission Spectroscopy (IES) [1-2]. The major advantages of IES are that the samples are measured insitu at the elevated temperature and IES requires no sample treatment other than making the mineral sample of submicron particle size. This technique overcomes the difficulties associated with the use of absorbance techniques, which in turn requires the use of pressed potassium bromide pellets or liquid parafin mulls (but these cannot be heated). Further the technique removes the difficulties of heating the sample to some higher temperatures and quenching to room temperature before absorption measurements. IES measures the thermal transformation process as it is actually taking place. In this paper we report the study of the thermal transformations of minerals and modified minerals using this IES technique.
Theory
Normally when using dispersive infrared spectroscopy, the infrared radiation is split into two beams, the reference and the measuring beam. The resultant absorbance is the negative logarithm of the ratio of the measured and reference intensities. Fourier transform instrument is, in essence, a single beam device hence the background is measured as a single beam and the measured spectrum ratioed to this background spectrum. In Fourier transform infrared emission spectroscopy the emission spectrum is actually this single beam reference or background spectrum, modified by the instrument response function. Emission spectra are obtained by ratioing the emission of the sample to that of a reference usually a black body source which emits a continuum of radiation according to Planck’s law. Real samples will always emit less energy than that of the blackbody. Consequently for any given temperature, the ratio of the energy emitted by the sample to that of the blackbody at any given wavelength is the emissivity.
Experimental
Infrared Emission Spectroscopy
FTIR emission spectroscopy was carried out on a Digilab FTS-60A spectrometer, which was modified by replacing the IR source with an emission cell. A description of the cell and principles of the emission experiment have been published elsewhere [1]. Approximately 0.2 mg of the inorganic material was spread as a thin layer on a 6 mm diameter platinum surface and held in an inert atmosphere within a nitrogen-purged cell during heating. The infrared emission cell consists of a modified atomic absorption graphite rod furnace, which is driven by a thyristor-controlled AC power supply capable of delivering up to 150 amps at 12 volts. A platinum disk acts as a hot plate to heat the sample and is placed on the graphite rod. An insulated 125-m m type R thermocouple was embedded inside the platinum plate in such a way that the thermocouple junction was <0.2 mm below the surface of the platinum. Temperature control of ± 2°C at the operating temperature of the sample was achieved by using an Eurotherm Model 808 proportional temperature controller, coupled to the thermocouple.
The design of the IES facility is based on an off axis paraboloidal mirror with a focal length of 25 mm mounted above the heater capturing the infrared radiation and directing it into the spectrometer. The assembly of the heating block, and platinum hot plate is located such that the surface of the platinum is slightly above the focal point of the off axis paraboloidal mirror. By this means approximately a 3 mm diameter area is sampled by the spectrometer. The spectrometer was modified by the removal of the source assembly and the mounting of a gold-coated mirror, which was drilled through the centre to allow the passage of the laser beam. The purpose of this hole is to allow the passage of the HeNe laser beam for optical alignment of the spectrometer. The mirror was mounted at 45°, which enables the IR radiation to be directed into the FTIR spectrometer.
In the normal course of events, three sets of spectra are obtained: firstly the black body radiation over the temperature range selected at the various temperatures, secondly the platinum plate radiation is obtained at the same temperatures and thirdly the spectra from the platinum plate covered with the sample. Normally only one set of black body and platinum radiation is required for an uninterrupted series of experiments. The emittance spectrum at a particular temperature was calculated by subtraction of the single beam spectrum of the platinum backplate from that of the platinum + sample, and the result ratioed to the single beam spectrum of an approximate blackbody (graphite). This spectral manipulation is carried out after all the spectral data has been collected.
The emission spectra were collected at intervals of for example, 50°C over the range 200 – 750 °C. The time between scans (while the temperature was raised to the next hold point) was approximately 100 seconds. It was considered that this was sufficient time for the heating block and the powdered sample to reach temperature equilibrium. The spectra were acquired by coaddition of 64 scans for the whole temperature range (approximate scanning time 45 seconds), with a nominal resolution of 4 cm-1. Good quality spectra can be obtained providing the sample thickness is not too large. If too large a sample is used then the spectra become difficult to interpret because of the presence of combination and overtone bands. Self-absorption can also result from having too thick a sample.
Discussion
The single-beam emission spectra of the graphite background spectra obtained between 200 and 700 ° C are shown in Figure 1. It is noted that as expected, there is an increase in the instrument response to higher frequencies as the temperature is raised. The greater thermal energy upon raising the temperature results in significantly increased intensities. Figure 2 illustrates the thermal response from the platinum plate. The intensity response for the platinum is less than that of the graphite.
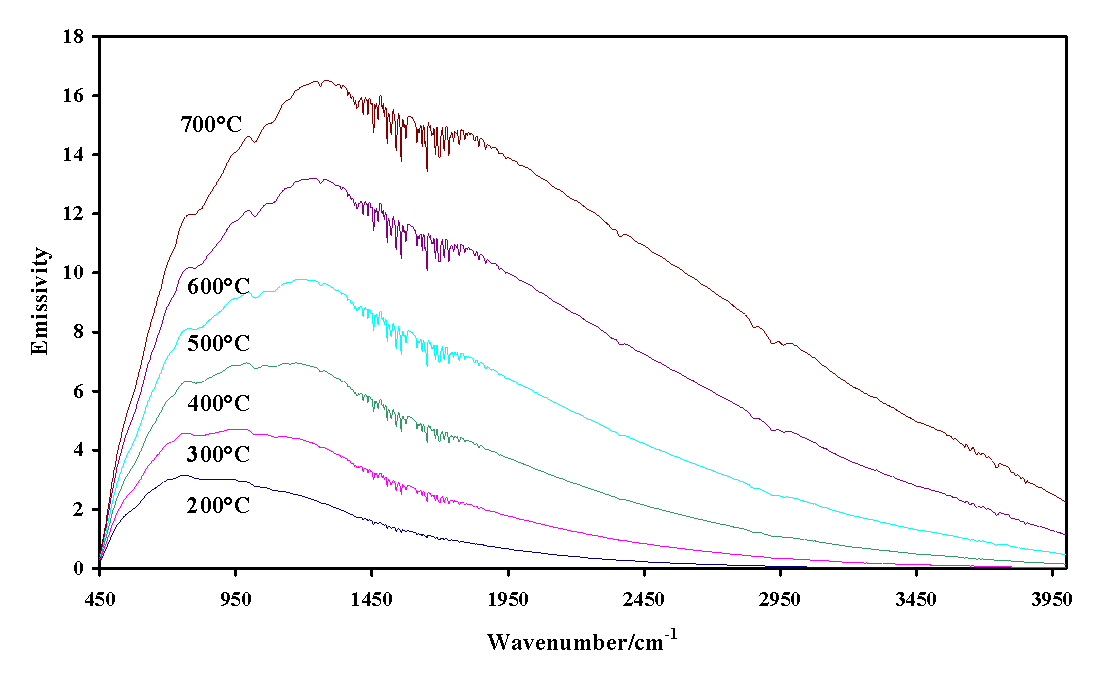
Figure 1. Infrared emission spectra of graphite from 200 to 700 ° C
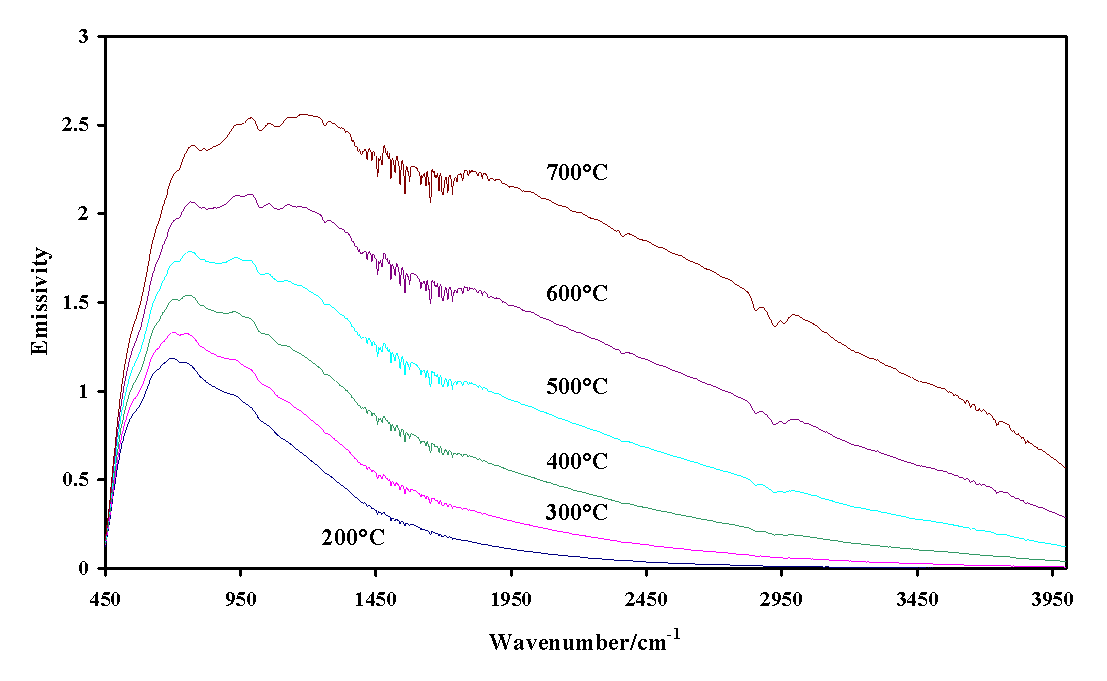
Figure 2. Infrared emission spectra of platinum from 200 to 700 ° C
The emission spectra of some naturally occurring and synthetic minerals
Figure 3 shows a comparison of the infrared absorption spectra (in red) and the 200° C infrared emission spectra (in blue) of hectorites. The emission spectra show the presence of water vapour in the spectra despite extensive purging, as water is being lost as the hectorite is being heated. The SHCa-1 sample is the Clay Minerals standard, which has been calcium exchanged. The spectra labelled ‘Hector’ are from an unknown sample. What is clearly evident is that absorption and emission spectra are identical. The emission spectra have the atmospheric water vapour spectra superimposed upon the emission spectra. Hectorite is a trioctahedral magnesium smectite clay mineral of the formula [Caz(Mg6-zLi2z)(Si8)O20(F,OH)4]. The lettering in Figure 3 shows some of the different spectral features of this mineral. The bands labelled A show the hydroxyl stretching bands of the hectorite which are retained to quite high temperatures. B shows organic impurities in the mineral. C demarks the presence of carbonates in the mineral. D is the SiO stretching bands of the hectorite. E is the hydroxyl deformation vibration corresponding with the hydroxyl stretching bands at A. The infrared absorption spectra of the hectorites are identical to the emission spectra. These spectra enable the molecular structure of the hectorite to be obtained.
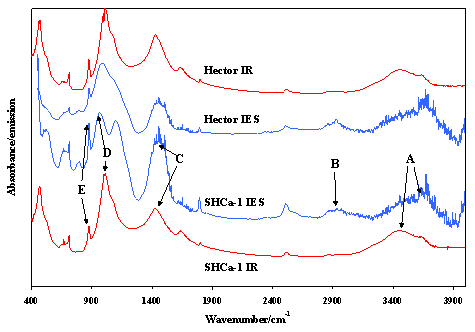
Figure 3. Infrared absorption and emission spectra of a standard and an unknown hectorite
One of the obvious advantages of infrared emission spectra is the ability to obtain a suite of temperature-consecutive spectra of a sample in situ at the elevated temperatures. Figure 4 clearly shows such a suite, exemplified by a pillared hectorite.
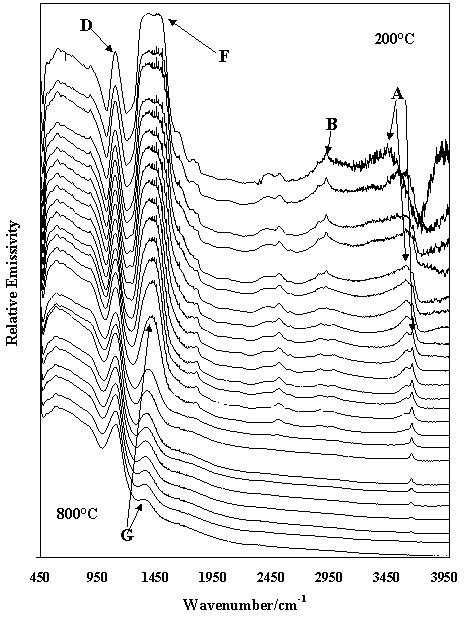
Figure 4. Infrared emission spectra of the Al13 pillared hectorite in the 200 ° to 800° C at 25 ° C intervals
Pillaring is a process of inserting pillars of for example Al13 or [AlO4Al12(OH)24 (H2O) 12] 7+ (known as the Keggin structure) between the layers of the hectorite to generate a new material [3-4]. The pillar is fixed in the layers by thermal treatment at around 600 ° C where the Al13 is transformed into Al2O3. Bands labelled A show the hydroxyl-stretching region of the pillared hectorite. The spectra in this region are different from those in Figure 3 as the water in the Keggin type (Al13) structure is also present. Water OH stretching frequencies are observed around 3350 cm-1 and these bands are lost by 250° C. The OH stretching frequencies of the Keggin structure are retained until 600 ° C and of the hectorite are retained until 775 ° C. B type bands are due to the presence of organics in the clay. The intensity of these bands approaches zero by 550 ° C. D demarks the SiO stretching vibrations and changes in these bands may be observed as the alumina pillar reacts with the siloxane surface. Fdemarks the (AlO4Al12(OH)24) pillar after insertion between the hectorite layers. Changes in the molecular structure are observed as thermal treatment takes place. Eventually the alumina pillar is inserted between the hectorite ones as observed by the spectrum demarked by G. The presence of the pillar and the changes to the molecular structure of the pillar may be observed by IES spectra in the 1400-1450 cm-1 region. The set of spectra also shows the dehydroxylation of the hectorite. Changes in the SiO stretching bands are also observed upon thermal treatment and its is suggested that as the pillar is locked into the structure, a reaction between the alumina pillar and the siloxane surface occurs. Thus IES technique allows the identification of new inorganic materials and the changes in the molecular structure of these materials as thermal transformations occur.
Hydrotalcites often known as LDH’s or double-layered hydroxides are anionic clays found in nature but more often synthesised as precursors for catalysts. The difference between the normal clay minerals and the hydrotalcites is that the clay layer surface charge is positive and consequently this layer charge is counterbalanced by anions such as carbonates or nitrate ions. Clays such as smectites have negative layer charges which is counterbalanced by the positive charge of cations such as Ca++, K+ or Na+. Figure 5 illustrates the low frequency region of a synthetic Ni hydrotalcite (Ni6Al2(OH)16 CO3.nH2O) and the set of spectra over the 200 to 750 ° C temperature range. A demarks the hydroxyl stretching region and changes in this region as thermal transformation occurs. B shows the presence of organics in the spectra. C shows the presence of the interlayer carbonate in the structure. D demarks the AlO bands. The bands observed at near 2100 cm-1 is probably the overtone of this band. Clearly the spectra show the changes in the molecular structure of the hydrotalcite as it is transformed into a Ni spinel and NiO.
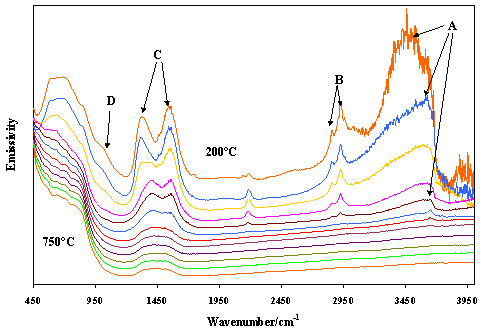
Figure 5. Infrared emission spectra of Ni-hydrotalcite in the temperature range 200 ° to 750 ° C at 50 ° C intervals.
Industrial Applications
Infrared emission spectroscopy has the potential for wide applications in industry, such as in process control and quality assurance. Providing the sample is at some elevated temperature compared to the detector temperature then a spectrum of the sample may be obtained. An important industry in Australia is the production of alumina, which depends on the mining of bauxite. Bauxite is predominantly composed of mixtures of gibbsite (Al(OH)3)and boehmite (AlO(OH)) in an approximate ratio of say 70:30. Figures 6 and 7 illustrate the thermal transformations of gibbsite and boehmite [5-6]. Of the many changes in molecular structure that may be observed is the difference in the dehydroxylation temperatures of the gibbsite and boehmite. Gibbsite dehydroxylates at 225-250 °C whereas boehmite does so at 400-450 ° C.

Figure 6. Infrared emission spectra of gibbsite over the temperature range 200° to 750 ° C at 50 ° C intervals.
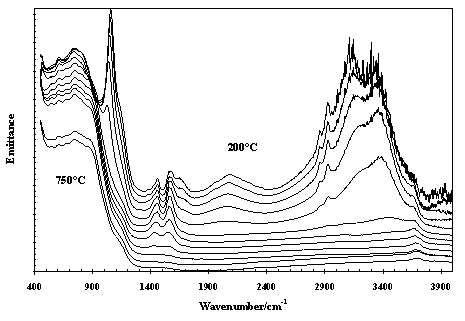
Figure 7. Infrared emission spectra of boehmite over the temperature range 200° to 750 ° C at 50 ° C intervals
Kaolinites and the other polytype halloysite, which can be obtained in high purity in nature, are often used in industrial applications: for example kaolinite is used as a paper coater and paper filler. It is also used as a filler in plastics and calcined kaolinite is used as a whitener in paints. Halloysite differs from kaolinite in that the structures are curved or rolled as opposed to the planar structure of kaolinite. Halloysite is often used as component for the manufacture of ceramics. Figure 8 shows the changes in the molecular structure of the kaolin as calcination takes place. A shows the hydroxyl stretching bands of the kaolin and the loss of intensity in these bands upon thermal treatment. B demarks the SiO stretching vibrations and the changes in these bands as dehydroxylation occurs. C shows the hydroxyl deformation vibration and the loss of intensity in this band as dehydroxylation occurs. No changes in the structure of the kaolinite occur until after the dehydroxylation has taken place. Changes in the low frequency region of the spectrum are also observed. What is obvious is that the application of the IES technique to the study of the thermal transformation of minerals is endless. The technique allows the study of processes such as dehydration, dehydroxylation, phase transformations, adsorption and thermal desorption and even oxidation and reduction processes to be studied. Further spectra may also be obtained not only in a heating cycle but also in the cooling as well. Further the technique allows for the possibility of undertaking kinetic experiments.
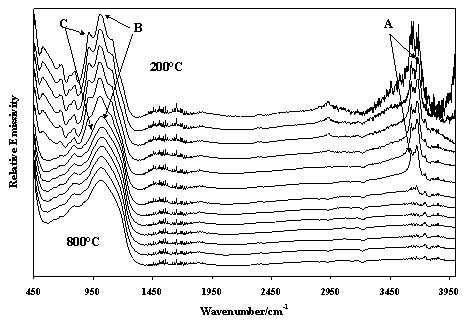
Figure 8. Infrared emission spectra of halloysite over the temperature range 200° to 750 ° C at 50 ° C intervals.
Advantages and disadvantages of the IES technique.
The major advantage of the IES technique is the ability to obtain spectra at the elevated temperatures in situ. This avoids the problems associated with the use of KBr pellets and heating of the pellets to elevated temperatures where reactions between the KBr and the for example clay may occur. The experiment whereby a sample is heated to an elevated temperature and then rapidly cooled to ambient is also not suitable as any reversible phase changes would not be observed. It has been shown that emission spectra are the same as the absorption spectra although some intensity differences in the bands are observed. Bands are generally broader in the emission spectra. Selection rules for emission and absorption spectroscopy are identical. Another major advantage is that the hot sample itself is the source and so no instrumental thermal source is required.
One difficulty of the technique is the inability to use thick samples. Emission spectroscopy is subject to artefacts and spectral features such as self-absorption, effects of sample thickness, non-uniformity of temperature and refractive index and reflectivity changes for strong bands. The technique depends upon having a thin film or fine dispersion of a powdered sample on the platinum plate. If too thick a sample is used then several problems may arise: firstly the appearance of combination and overtone bands. Such a difficulty may be overcome by reducing the amount of sample or by making the film thinner. Other high temperature infrared spectroscopic techniques such as the high temperature DRIFT spectroscopic method and transmission techniques using a Yates cell are available. However, the IES technique offers the advantage of greater signal intensity combined.
Acknowledgments
The financial and infra-structural support of the Queensland University of Technology, Centre for Instrumental and Developmental Chemistry is gratefully acknowledged.
References
- A.M.Vassallo, P.A.Cole-Clarke, L.S.K.Pang, A.Palmisano. J. Appl. Spectrosc., 46, 73, (1992).
- Ray L. Frost and Anthony M. Vassallo, Clays and Clay Minerals, 44, 635 (1996).
- J. Theo Kloprogge, R.L. Fry and R.L Frost. Journal of Catalysis, 184, 157 (1999).
- J. Theo Kloprogge, R.L. Fry and R.L Frost. The Analyst, 124, 381 (1999).
- Ray L. Frost, J.T. Kloprogge, S.C. Russell and J. Szetu. Applied Spectroscopy 53, 423 (1999).
- Ray L. Frost, J.T. Kloprogge, S.C. Russell and J. Szetu. Applied spectroscopy 53, 570 (1999).
Received 13th July 1999, accepted May 2nd 2000
REF: Ray L. Frost, Huada Ruan and J. Theo Kloprogge,
Internet J. Vib. Spec.[www.irdg.org/ijvs] 4, 1, 5 (2000)