Raman Spectroscopy in Pharmaceutical Analysis
Patrick. J. Hendra
Dept of Chemistry
University of Southampton
Highfield
Southampton
SO17 1BJ
United Kingdom
Email to: ijvs [at] soton.ac.uk
Abstract
Raman spectroscopy is of potential attraction as a tool in pharmaceutical analysis. It has been demonstrated that some analyses of complete formulations, audits on polymorphs and some on-line and at-line monitoring are all feasible but the lack of widespread acceptance of the method in routine general analysis has meant that the pharmaceutical regularities have been reluctant to licence Raman analytical procedures.
The paper describes the Raman method as it currently applies to pharmaceutical analysis and was presented as the opening paper in a one-day forum on Raman Spectroscopy organised by the the Royal Pharmaceutical Society of Great Britain on 18th February 1999 in London.
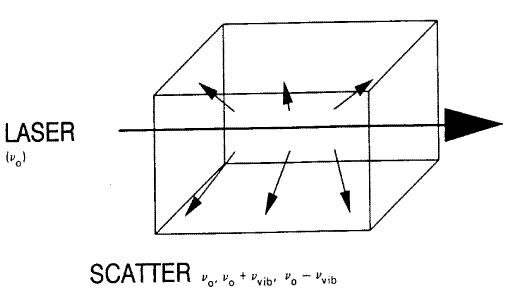
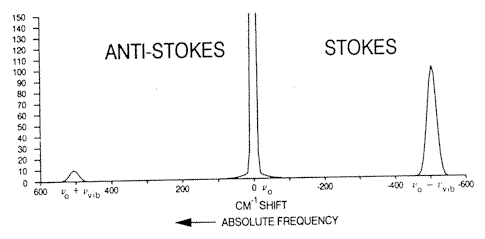
Figure 1. When an intense monochromatic light source of frequency vo irradiates a sample, the light scattered contains frequency components vo’vo + vvib and vo – vvib. In the case of liquid chlorine, the Stokes and anti-Stokes Raman bands are shifted by 505cm-1 from the excitation frequency. If the spectrum is excited with an argon ion laser vo = 19436cm-1, vo+ vvib = 19941cm-1 (anti-Stokes) and vo – vvib = 18931cm-1(Stokes).
The Stokes :anti-Stokes ratio intensity is approximately 10:1. [Reproduced from Ref 2.]
If a specimen is illuminated by a monochromatic source – these days invariably a laser – the scattered light will consist of light at the frequency of the source Vo ± Vvibrational. If the source has a wavelength of 1 micron i.e. 10,000cm-1 and the sample vibrates at 500cm-1 then two incredibly weak scattering ‘sidebands’ will occur at 9500 and 10,500cm-1. These bands are the Raman effect predicted in 1923 by Smekal and discovered five years later by Sir C.V. Raman.[1]Molecular vibrations give rise to both mid infrared absorptions (i.e. their frequencies lie between 200 & 3700cm-1 (50 – 2.6µm wavelength)) but also Raman scattering. Raman scattering is an inelastic scattering process defined in Figure 1.
Thus, Raman Spectroscopy is an alternative method for us to record the vibrational characteristics of a sample in the full knowledge that this very vibrational behaviour constitutes an excellent fingerprint for the specimen under analysis.[2]
In Figure 2, I show the combined mid infrared and the Raman spectra of a specimen. You will notice that to compare the two spectra, infrared at the top Raman at the bottom, the axes are different in each case: in the infrared we plot transmission against the spectroscopic frequency. In the Raman, the plot is scattering intensity vs wave-number shift from the wave-number value of the laser. In this way, the two measurements of the vibrational properties of our molecule are directly comparable.
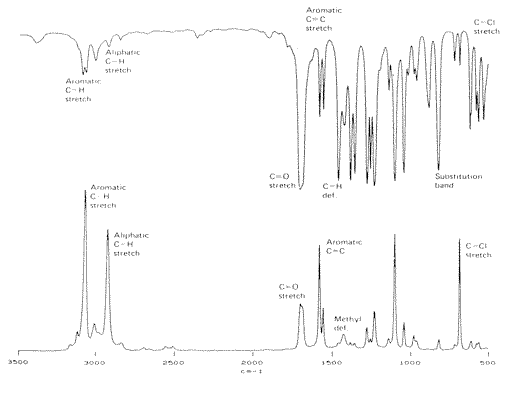
Figure 2. The infrared and Raman spectra of 2,5-Dichloroacetophenone
What do we see? The overall spectrum is similar in the Raman and in the infrared, but the shapes of the two spectra are totally different. If both techniques are measuring the same set of molecular properties this seems odd.
In infrared spectroscopy, we find that resonance occurs between the electromagnetic radiation bathing the sample when a molecular vibration contorts in such a way that the molecular dipole periodically varies. If it doesn’t – no resonance – no absorption of the mid infrared radiation. Raman scatter occurs when the polarizability of the molecule varies as it vibrates. The polarizability is best described to students as the ease of which electrons can be induced to slosh around a molecule as a field is applied. This polarizability (the sloshability!) can vary as a molecule contorts and in some vibrations this does indeed occur. Hence, some vibrational motions give rise to infrared absorption, some to Raman bands and some to both but in this latter case one would not expect the intensities of the interactions to be the same in each spectrum.
One of those dreadful rules of thumb which work but are always letting us down can now be enunciated. Symmetrical vibrations tend to give strong Raman bands, asymmetric ones give prominent absorptions in the mid infrared. Also the “group frequencies” are dissimilar.
Group frequencies are an incredibly old concept going back to Coblentz at the turn of the century (1900 not 2000!). Specific chemical functional groups often have a vibrational frequency associated with the group, which enables infrared users to carry out molecular diagnosis. Excellent examples are
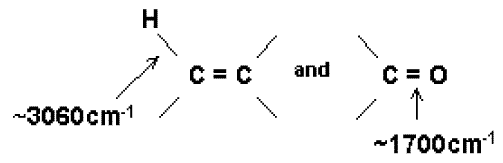
Raman spectroscopists have their own group frequencies but they are completely different from those of the infrareders. Thus the
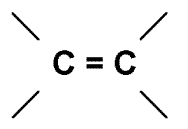
group always generates a strong Raman scattering line near 1680cm-1 shift
(shorthand D V » 1680cm-1).
To summarise: we can examine the vibrational characteristics of a molecule by two complimentary techniques – mid infrared absorption and Raman scattering. Obviously, to do the analytical job properly one ought to record both but hardly anyone does – or rather did! A simple description of the Raman effect will be found in Ref 3.
FT-Raman Spectroscopy
Raman spectroscopy has, since it’s discovery been dogged by fluorescence. If a sample is only slightly fluorescent or if it is contaminated with a trace of a fluorescer, the fluorescence will be stronger than the very weak Raman lines and inundate them. As a result, Raman spectroscopy has failed to attract analysts. What is the point of applying a method where impure or degraded specimens are inaccessible? As a result, there has been a tendency to use infrared along with nmr and mass spectroscopy as front-line analytical methods.
Late in the 1980s, commercial Raman systems began to appear that used near infrared laser sources (hence avoiding the fluorescence problem)[4,5]. An even more exciting development was that the new Raman facilities were offered as accessories on FTIR instruments. These F-T Raman accessories are now offered by all of the leading manufacturers and thousands are now in service. The combined FTIR/FT Raman system provides the analyst with a complete vibrational spectroscopic facility all in one instrument.
Comparison of FT-IR and FT-Raman
The Raman experiment is a scattering process so the experiment involves hitting a sample with intense monochromatic light, collecting and analysing the scatter. To compare the two processes, infrared absorption and Raman scatter study Figure 3. On the left, I draw the optical system favoured by all the various producers except Nicolet of America. This manufacturer prefers to collect the scattered light with a gold surfaced mirror rather than a lens but the principle is exactly the same – laser light onto the sample – collect the scattered light in the reverse direction… On the right of Figure 3, I draw a simple transmission experiment and quote the Beer Lambert Law. Clearly the amount of radiation absorbed can be controlled both by varying the thickness of the sample and the concentration of the analyte. Under the left-hand diagram you will see the Raman equivalent of the Beer Lambert Law. Note that the intensity can only be varied by changing the concentration of the analyte and the laser power. Sample thickness is not a useful variable but the mean volume irradiated is of importance.
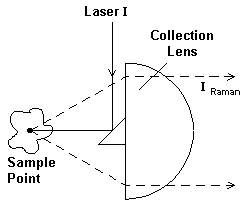
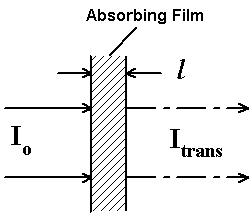
Figure 3. Backscatter Raman
IRaman= Il.k.X.c
X= Raman scattering cross-section of a particular band in a given substance.
C= corre. of scatterer
Il = Intensity of laser
Itrans/Io = e-ecl Beer lambert Law
e = extinction coeff. of each band
C= conc. of absorber
Let us then compare and contrast FTIR and FT Raman
| Infrared absorption FTIR | Raman Scatter FTR |
| Has its own group frequencies | Has its own group frequencies |
| Must use infrared transmissive optical materials like KBr or ZnSe | Uses glass or quartz as optical materials hence bottles and tubes are OK. |
| Needs a really thin film for transmission measurements (~30µm typical) | No sample preparation needed. If the sample fits into the instrument. |
| No fluorescence problem | Even with nir laser sources occasional samples fluoresce |
| Beer-Lambert law enables users to measure traces – thick samples are involved. Hence Excellent Sensitivity |
No real equivalent of Beer-Lambert law. Increasing laser power to increase sensitivity is possible but raises the possibility of ‘burning’ the sample.Hence Poor Sensitivity |
| Even allowing for sample preparation time – fast ~6/hrSkill/ experience essential in preparing samples | As no sample preparation – very fast ~ 15/hrNo skill required (almost completely Undergraduate proof!) |
| Inherently safe | Laser is powerful and invisible (Class IVb) hence very dangerous\manufacturers interlock the sample chamber lid and laser |
| Optical fibre probes for ‘off bench’ remote application available but are short (½-1m) and very inefficient. | Nir radiation passes efficiently through silica fibres hence F-O probes are used. |
| Quantitative Pharmaceutical AnalysisHas value but dilute aqueous solutions not attractive | Major components particularly in solids – good quantitative accuracy but there are cases where this is not so.Dilute aqueous solutions NOT accessible. |
So the obvious question arises – why should I consider using Raman as an analytical procedure when I already have available a compete suite of more conventional techniques? The answer is that for specific analyses where Raman works well, the ability to record spectra from unprepared samples in glass vessels or through glass windows and the possibility of operating remotely are very attractive. The experiment is entirely open-ended – the laser impinges on the sample and the scattered light is collected in the reverse direction so sample size and shape are irrelevant. Now, the Raman scattering of some components is selective i.e. scattering from one compound can be much more intense than from others and this can be exploited – many actives give much stronger spectra than excipients and this can be used to make quantitative measurements even when the active is present at low levels. Frequently, different polymorphs or hydrates give really different Raman spectra hence production measurements and control or formulation monitoring and shelf life studies are all in prospect. In addition, Raman provides a laboratory analytical tool for the synthetic chemist complimenting FTIR, NMR and Mass Spectrometry.
FT Raman vs CCD systems
The account above extols the virtues of FT-Raman – near infrared sources and an accessory on an FTIR. There is an alternative method of recording spectra, which is much closer to the experiment carried out by Raman himself in 1928. Rather than using the Fourier Transform/Interferometer as the basis one buys a dedicated Raman instrument which incorporates a Charge Coupled Device # detector. Because these detectors operate in the visible and very deep red, these instruments incorporate a VISIBLE laser.
# An up-market version of the detector in your Camcorder.
The latest CCD detectors are incredibly sensitive and are used in the most advantageous way in the instruments on offer. As a result, they have much more sensitivity than their FT Raman competitors.[6] “Much more” is 100 to 1000x so it is really worthwhile. In Figure 4, I show the block diagram of a typical CCD based instrument. Several points are noteworthy – these instruments very frequently use a modified conventional visible microscope as the sample system. They will also work through optical fibres so these visible laser CCD systems are the best Raman machines on offer WHERE THEY ARE APPLICABLE. Thus, it is worth considering briefly the types of sample where Raman analysis can be useful, comparing CCD and FT machines.
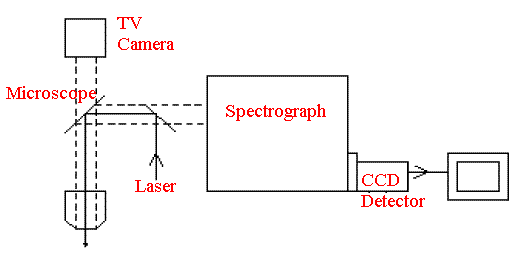
Figure 4.
Sample types in Pharmaceutical Analysis
- Analytical support to research and forensic departments – the breadth of analytical requirements makes it inevitable that fluorescence will be a real nuisance ® Recommend F-T Raman (nir laser)
- Manufacturing support and on-line analyses e.g. the (crystal) monitoring modification of crystallites as they precipitate. Homogeneity studies. In theses cases ‘pure’ compounds are involved and hence fluorescence tends to be less of a problem. Recommend CCD based instruments (vis. laser)
- Examination of formulations – quantitative measurements on actives. Usually these materials are complex and frequently dyed and hence fluoresce. Recommend F-T Raman (nir laser)
- The analyses of relatively dilute and very dilute solutions. This particularly applies to solutions where the solutions are poor scatterers e.g. saturated bio derived CHO and N atom compounds.
- Raman microscopy. Although FT Raman microscopes exist they are completely outclassed by the CCD based systems.[7]
However the real point is that the individual analysis will, in effect, define the approach to be adopted. Thus, well-endowed laboratories have both systems.
To give you a feel for the quality of good Raman spectra here is an example.
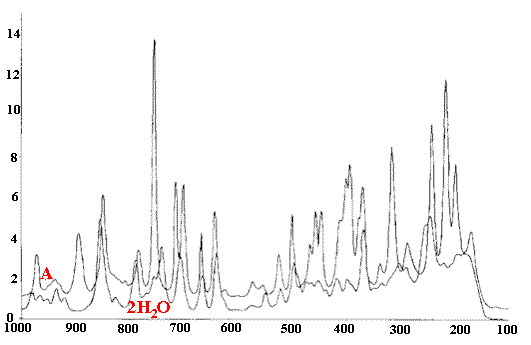
Figure 5. Norfloxacine anhydrous (A) / dihydrate overlayed (2H2O)
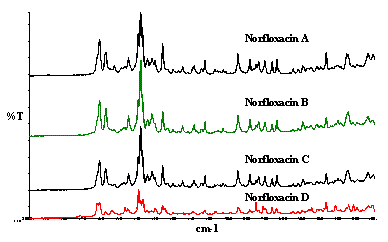
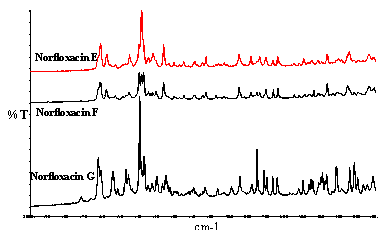
Figure 6. Examples of polymorphs of Norfloxacin.
Some specimens if not all are mixtures
References
- C.V. Raman, 1928.
- Fateley et al, The Handbook of Infrared and Raman Characteristics Frequencies of Organic Materials, see Spectroscopists Bookshelf, Item 15
- P.J Hendra, Internet J.Vib.Spect (www.ijvs.com)
- D. B.Chase, J. Am. Chem.Soc., 108, 7485, (1986).
- P.J. Hendra, C.Jones and G.Warnes. Fourier Transform Raman Spectroscopy, Ellis Howard, Chichester Press UK, (1991).
- M.J. Pelletier, Internet J.Vib.Spect (www.ijvs.com) 1, 3, section1, (1997).
- B. Cooke, Internet J.Vib. Spect (www.ijvs.com) 2, 4, 2 (1998).
Received 9th February 1999, received in revised format 12th February 1999, accepted 12th February 1999