FT-NIR Spectroscopy in the Ink and Paint Industry
Eszter Trenka and Joachim Oelichmann
Büchi Labortechnik AG
Meierseggstrass 40
CH-9230 Flawil
Switzerland
Introduction
Colour is one of the most demonstrative features of all kinds of goods. In most cases organic colored pigments are used to achieve the desired appearance. However pigments influence much more properties than just the colour, therefore the definition and maintenance of quality standards of the intermediates and raw materials used in pigment synthesis and paint production is crucial. The chemicals used range from organic fine chemicals for synthesis to products of inorganic basic chemistry and to special surface active agents, surfactants and resins.
These requirements lead to an increasing demand for quality control in the whole branch of industry. According to the extensive norms (ISO, TQM) the tests should include the various stages of production from raw material to end product. The necessity of many measurements cause increasing costs. Therefore a fast, precise, simple and cost saving analytical technique is needed.
FT-NIR spectroscopy is a universal and efficient analytical technique for substance identification and quality assurance. The main advantages are its speed, dependability and minimized sample preparation required, which make it the best qualified method.
Today quality control by NIR spectroscopy is an integral part of the process during the production of inks and paints. It is efficient and motivating for the employee in the production to control various chemical and physical parameters by himself and to introduce necessary corrections. The aim of the tests is to maintain the quality of the end products.
Not only the producer of inks and paints but also the producer of the raw materials need to perform quality tests, which are not only restricted to simple physical parameters. Legal requirements and specifications of his customers must be fulfilled. The last member of the simplified “quality chain” is the user. Every member contributes to the quality of the final end product.
Near-Infrared spectroscopy
In the near-infrared range, in particular overtones and combinations of OH-, NH- and CH-vibrations can be observed. The most universal range is between 9`000 cm-1 and 4`000 cm-1, which delivers good spectra of most organic substances and is widely applied. The band intensities rarely vary very much between different compounds. In any case, they are such that even without sample preparation measurements can be performed either in transmission or in diffuse reflectance using an optical fiber probe. In concentrated systems and due to strong overlapping of the absorption bands, the Lambert-Beer law is normally not applicable. These circumstances make it necessary to use somewhat more comprehensive algorithms for the interpretation of the spectra. The most commonly used chemometric methods are principal components analysis, principal component regression and partial least squares regression.
Controlling of raw materials
A lot of different raw materials are used during the production of inks and paints. The palette includes binders, solvents, pigments, fillers, additives and auxiliary materials. Normally only simple physical tests are performed or to some extent wet chemical methods like acid-, saponification- or iodine-values are determined. Especially the latter procedures are time consuming and need chemicals, which need to be bought, stored and disposed of. Therefore these tests are expensive.
NIR spectroscopy is a very good tool for replacing such wet chemical tests. It is possible to assure positive identification of incoming raw materials within seconds. The risk of using incorrect materials is drastically reduced. Practical experience demonstrates, that the price of non-conformance (using wrong or out-of-specification materials) is in the range of some percent of the sales volume, which can easily be reduced.
Production and end product control
During the last few years efforts have been undertaken in the industry to control process parameters that are critical for assuring the product quality. The conventional approach used to be to add a quality control stage after the process, which however had some drawbacks. If a production batch was rejected because of not-meeting the specifications, a series of costly corrective actions had to be taken. Today, instead, the process can be controlled so that all of the goods produced comply with the specifications.
Using NIR spectroscopy the shift worker can present a sample to the instrument and he can be told in a matter of seconds, what he has to do to ensure that everything will come out as it should be.
The goal of these measurements can be to identify raw materials or end products, to determine the concentrations or concentration ratios in blends or reaction feedstocks, or to establish the content and uniformity of content of formulations.
FT-NIR system
All spectra of the examples shown had been measured with a NIRVis or NIRFlex N-400 FT-NIR spectrometer It is based on a polarization interferometer with two quartz wedges [ 1 ] and features high stability and robustness, which is essential especially for the use in harsh environments. Therefore it is possible to use the instrument not only in the laboratory but directly on the platform or in the production area as well.
For the calibrations the chemometric software package NIRCal (NIR Calibration) had been used. It supports material identification and qualitative or quantitative applications as well. NIRCal uses chemometric algorithms (PCR, PLS, MLR, Cluster Analysis) for efficient data compression and evaluation [ 2 ]. It is strongly dedicated to the needs of the operator and easy to use. During routine use the operator is forced to follow a defined procedure and all inputs can be done via a barcode reader and remote control.
For method development there are comprehensive and flexible tools to select the best model for each calibration. In order to optimize a method, a large number of different parameters are available to the user, which can be combined in any sequence and in any number. Until now comprehensive chemometric experience and and lot of time had been necessary for the development of ideal calibrations.
With the NIRCal software the hurdles are greatly simplified. The implemented NIRCal Wizards guides the user step-by-step from the registration of the spectra to calibration and validation of the method. The Calibration Wizard especially completely relieves the user of time and money consuming tasks. The user has simply to answer some questions regarding the used sampling option, the nature of the samples to be investigated, the desired calibration quality and type (qualitative or quantitative). The Wizard then uses its vast knowledge base, calculates the possible calibrations, and automatically documents all results obtained. The various calibrations can be investigated and judged in the NIR-Explorer using the Q-value – an objective measure of the quality of the calibrations. Main advantages offered by this approach are that the user`s chemometric knowledge will affect the results only slightly, and that the results therefore become reproducible. The new calibration tool offers a particularly large potential for reducing calibration effort and costs [ 3 ].
Applications
In the following some applications are described. They do not represent the complete possibilities but only give some typical examples. More examples can be found under www.ft-nir.com.
Identity control of incoming raw materials
The identification and quality control of incoming materials is a straightforward application, which can be performed directly on the platform without any further time consuming tests.Organic solvents have different functional groups like alkyl-, hydroxyl-, aromatic CH-links or others. These show characteristic absorption bands, which support the automatic identification.
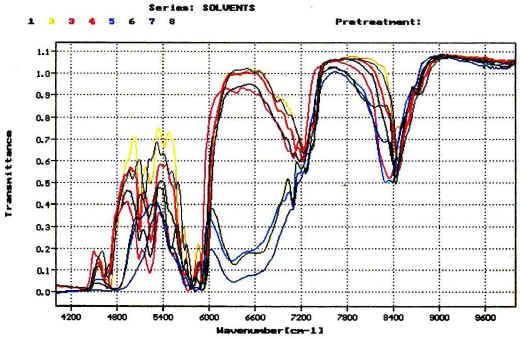
Figure 1. NIR spectra of different solvents used
frequently in the ink & paint industry.
Figure 1 shows the spectra of butyl acetate, acetone, cyclohexanone, methyl ethyl ketone, 1-butanol, 2-propanol, ethanol and ethyl acetate. All solvents are commonly used in the ink and paint industry. It can easily be seen that the spectra are sufficiently different for the establishment of an identification.
In this example a transmittance fiber optic probe had been used. With such an sampling option it is possible to do the measurements directly in a barrel or even in the tank lorry.

Figure 2. Measuring liquids directly in the original
containers using a transmittance fibre optic probe.
It features a solid construction with integrated remote control for measurements even of highly corrosive liquids. With the remote control it is possible to start the measurement directly without the need of operating a PC. Additionally the result (OK or NOT OK) is indicated at the device.The pathlength (1.5 mm) of the probe is optimized for the investigation of various liquids.
Quality control of resins
Resins are complex polymers which often are dissolved in different solvents. Today water based systems are increasingly used. The quality control of such systems by classical chemical methods is time consuming and rather expensive. With NIR spectroscopy this can be done very easily and fast. The example shown below demonstrates how to check the quality of the phenolic resin. Bad qualities can be rejected at a very early stage of the production.
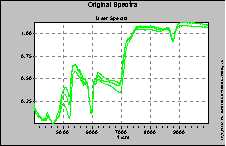
Figure 3. Spectra of phenolic resins.
The measurements shown below had been performed in the transflectance mode. This can be done with a special attachment to the standard reflectance fiber optic probe, which is mounted directly on the reflectance probe head.
With this sampling option there is a small gap between the end of the fiber optic probe and a diffuse reflector. The pathlength can be optimized for the application. This gap is filled with the sample. The radiation passes the sample, is reflected and passes the sample again. In this way a transmission measurement is performed using the indirection of reflectance.
Quantitative analysis of resins
Additionally it is not only possible to differentiate between various qualities but also to quantify the components of resins. As an example the quantitative determination of formaldehyde, phenol and water in a phenolic resin is given. The measurements had been done with the transmission fiber optic probe described already above. Only a few scans had been accumulated, which means that the result is obtained within less than 30 seconds only. The standard error of prediction (SEP) is about 1.5. This shows that the amount of the components can be determined with high accuracy.
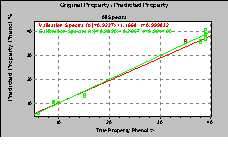
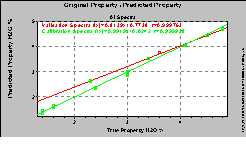
(a) (b)
Figure 4. Calibration and validation results for the
estimation of phenol (a) and water (b) content.
Determination of NCO content
As mentioned above classical analytical methods often involve wet chemical procedures. Titration is a common technique for the determination of NCO content. NIR spectroscopy is an indirect method, which means that a calibration is needed before it can be used for the analysis of unknown samples. The standards used must be well characterized by classical analytical techniques.The uncertainties of these techniques are inherently transferred to the chemometric methods used. However the following example (Du Pont, Wuppertal) demonstrates that in fact it will deliver comparable results [ 4 ].
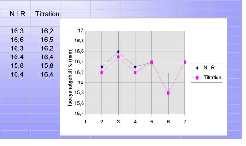
Figure 5. Comparison of NIR and titration results
for the estimation of NCO content.
Analysis of pigments
At the beginning it had been mentioned that pigments are very important for the quality of the paints. Therefore of course they are subject of an extensive quality control as well. The next example will address this segment.
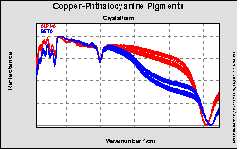
Figure 6. NIR spectra of copper phthalocyanine pigments.
Copper phthalocyanine pigments are often used as blue colour. They exist in two crystalline modifications, the so called alpha and beta forms. It is necessary to identify both forms, because of their different properties. In total 80 spectra of both forms had been measured in diffuse reflectance using the standard fiber optic probe described above. It is easily possible to set up a calibration for the differentiation of both forms. The following cluster plot demonstrates the excellent separation of both forms.

Figure 7. Cluster plot for the differentiation of alpha and beta
forms of Cu-phthalocyanine pigments.
Conclusion
FT-NIR spectroscopy allows both efficient identification and quantification for the quality assurance. It can be implemented at all stages of production and locations from the platform for incoming raw materials, the quality control laboratory to the production floor. The technique offers particular advantages and additionally offers fascinating prospects for online analysis.
References
- J. Knecht, Design and Application of a Near-Infrared Polarization Interferometer for Industrial Use, Microchimica Acta (Suppl.) 14, 769-771 (1997).
- H. Martens, T. Naes, Multivariate Calibration, John Wiley & Sons, New York, 1989.
- J. Ebert, R. Bossart, J. Grabinski, H. Kellerhals, Wizard for the Calibration of Büchi NIR Spectrometers, best@buchi Industrial 07 2001.
- N. Basener, NIR zur Qualitätskontrolle in der Lackindustrie, XXIV. FATIPEC Kongress, 8-11.June 1998, Interlaken, Switzerland.
REF: E. Trenka & J. Oelichmann, Int. J. Vib. Spect., [www.irdg.org/ijvs] 6, 3, 2 (2002)