The SERS Effect as a Tool for Studying Molecules Adsorbed on Semiconductor Surfaces
Lucia G. Quagliano
Consiglio Nazionale delle Ricerche (C.N.R.) – IMAI
Area della ricerca di Roma,
P.O.Box 10
00016 Monterotondo Scalo (Roma),
Italy
E.mail:quaglian [at] mlib.cnr.it
phone: +39-06-90672214
fax: +39-06-90672238
Abstract
We have demonstrated that adsorbed species on semiconductor surfaces can be detected by SERS spectroscopy by using two SERS-activation procedures. One consists of depositing on the semiconductor surface an Ag island film, the other based on the use of a chemical etching process. The observed SERS spectra of the adsorbed molecules are similar in both SERS-activation procedures. No Raman scattering could be detected from the GaAs surface without either the etching procedure or the Ag film because the vibrational spectra of molecules adsorbed on semiconductor surfaces were too weak to be detectable without an enhanced Raman cross section. These results are important from the SERS point of view because they show that semiconductors are capable of supporting the SERS effect.
Introduction
The Surface Enhanced Raman Scattering (SERS) effect is the enhancement of the Raman scattering cross section by several orders of magnitude for atomic and molecular species adsorbed on certain rough surfaces [1-3]. The experimental discovery of SERS in 1974 [4] fascinated many scientists since it offered the prospect of its possible application to surface characterization. Until the discovery of SERS, the low sensitivity of Raman spectroscopy had limited the applicability of this spectroscopy in surface research. However its potential in providing molecular information has yet to be realised. The SERS technique has not become widely used in surface science because it is essentially restricted to noble, alkali and some transition metal substrates [5].
We have demonstrated that species adsorbed onto semiconductors can be detected by SERS if two SERS-activation procedures are used. One consists of overlying the semiconductor surface with a discontinuous film of Ag [6-9], the other is based on the use of a chemical etching process [10]. We have observed the SERS spectra of tris (bipyridine) ruthenium(II), [Ru(bpy)3]2+ adsorbed on semiconductors. We chose this material as a test molecule because of its potential in photochemical energy conversion. The Raman spectrum of these molecules adsorbed onto the rough semiconductor surface prepared by anisotropic etching is similar to that obtained from a surface onto which Ag island films have been deposited.
Although the purpose of this work was strictly to demonstrate that semiconductors are capable of supporting SERS, our results allow us to say unequivocally that by using the “Ag overlayer method” the SERS signal arises from molecules directly adsorbed to the semiconductor surface. It should be noted that the technique for depositing Ag island film on semiconductors should could be very general and may be applicable to other non SERS-active materials and to a wide variety of physical environments.
Experimental
Silver slides and semi-insulating GaAs, InP and Si samples (wafers and epitaxial layers) were utilized. We used two methods for preparing “SERS-active” surfaces.
One, called the “Ag overlayer method”, is based on the application of a discontinuous layer of Ag on the surfaces of interest [8,9,11]. The optimum thickness of the silver layer to produce the maximum enhancement factor is 50-100 Å.
Silver films of 50 Å thickness were deposited by thermal evaporation under ultra-high vacuum at the very low evaporation rate of ~ 0.3 Å/s. This low rate is necessary because slowly formed film tends to form island structures in contrast with “fast” film where continuous formation occurs. With the aid of a mask, the Ag was deposited on a section of the samples allowing a comparison between the silvered and nonsilvered areas. The overlaid semiconductor wafers were then immersed in a solution of 2.5×10-2M [Ru(bpy)3]2+ in acetonitrile to provide the adsorbed layers we required. After at least 15 minutes, the wafers were taken from the solution and rinsed in acetonitrile to remove any material not strongly adsorbed to the surface. Raman scattering was then measured from several places inside and outside the Ag deposit, before and after immersion in the solution of ruthenium complex.
The other method used for the GaAs semiconductor samples involves an anisotropic etching [12,13] procedure. The semiconductor surface is etched with a solution containing concentrated HNO3 and [Ru(bpy)3]2+. In this case the adsorption of complex cations occurs during the etching process. Following this treatment the samples were rinsed in acetonitrile for several minutes and subjected to a short ultrasonic agitation in acetonitrile in order to remove nonadsorbed molecules.
Raman measurements were performed on the samples in air for ex situ spectra collection. The scattering light was analyzed with a Spex Triplemate spectrograph equipped with a cooled EG&G CCD. The excitation light sources were the 514.5 nm and the 457.9 nm lines of an Argon ion laser.
Results and Discussion
In figure 1 we compare the Raman spectra of [Ru(bpy)3]2+ molecule free in solution with that adsorbed on a silver substrate onto which an Ag island film has been deposited. The Raman spectrum of aqueous solutions of [Ru(bpy)3]2+has been obtained with the 457.9 nm laser line because fluorescence dominates the corresponding spectrum at 514.5 nm excitation. The Raman spectrum of the complex molecule was only observed on the Ag coated surface, indicating that there is an enhanced Raman cross-section caused by the Ag. Identical SERS spectra was observed from Ag island film deposited on a glass substrate. An interesting point to underline is that the Raman spectra of the molecule adsorbed on silver substrates are very similar to those of the free molecules in solution. This must indicate that the structural differences between the molecule free and adsorbed on silver are small.
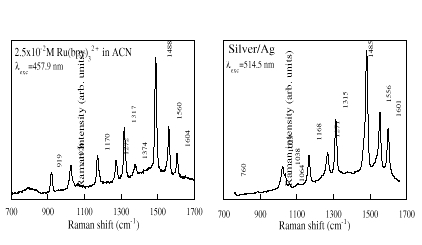
Figure 1. Comparisons of Raman spectra from [Ru(bpy)3]2+free in solution and adsorbed on silver/Ag system.
Opposite results were observed in the case of semiconductors as shown in Figure 2. In this picture we compare the Raman spectra of [Ru(bpy)3]2+ free in solution and that adsorbed onto Si, GaAs and InP surfaces coated with Ag island film. Laser light at 514.5 nm was used to avoid any resonance enhancement contribution from the molecule [14]. Raman scattering was measured from several places inside and outside the Ag deposit, before and after immersion in the solution. We could detect no Raman scattering from surfaces without the deposited Ag film i.e. the spectra of molecules adsorbed on semiconductors were too weak to be detectable without an enhanced Raman cross section.
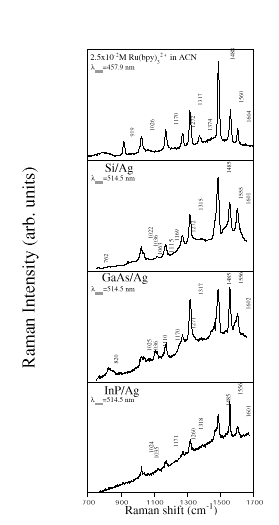
Figure 2. Raman spectra from [Ru(bpy)3]2+ free in solution and adsorbed on: Si/Ag, GaAs/Ag and InP/Ag systems.
Comparing the enhanced Raman spectra of the molecule we observed:
(1) the appearance of new features for the semiconductor/Ag systems, for instance the broad bands at ~820 and near 1110 cm-1 in the spectrum from GaAs. In our opinion these could well be due to new bonds between the molecule and the substrate, since they are absent in the Raman spectra before the immersion of the systems in the solution. After immersion they are present only in the measurements from places inside the Ag deposit.
(2) different absolute intensities due to, we think, to the distance of the molecule from the several substrates surfaces;
(3) different relative intensities of some lines indicating a change in the spatial orientation of the molecule with respect to the substrate;
(4) the appearance of a doublet, absent in the solution, at ~1025 cm-1 and ~1036 cm-1 ;
(5) slight wavenumber shifts with respect to free [Ru(bpy)3]2+ in solution.
Therefore, in the case of semiconductor substrates we have observed substrate dependent, linewidths and relative and absolute intensities of some lines depending on the kind of semiconductor. This behavior allows us to affirm with confidence that the SERS signals arises from the semiconductor surface and not from the Ag film.
In Figures 3a and 3b we show the Raman spectra of [Ru(bpy)3]2+ cations adsorbed onto two different GaAs surfaces, one coated with an Ag island film and the other subjected to a chemical etching process, respectively. Spectrum 3(b) shows the Raman spectrum obtained from a GaAs surface subjected to anisotropic etching containing [Ru(bpy)3]2+ solution and HNO3 as the etchant. In this case, the adsorption of molecules occurs during the etching process. The observed Raman spectrum was collected ex situ with the sample in air and is similar to spectrum 3(a). Evidently in this case the Raman scattering from the adsorbed molecules is enhanced, in fact we did not detect Raman scattering from GaAs samples immersed only in the [Ru(bpy)3]2+ solution.
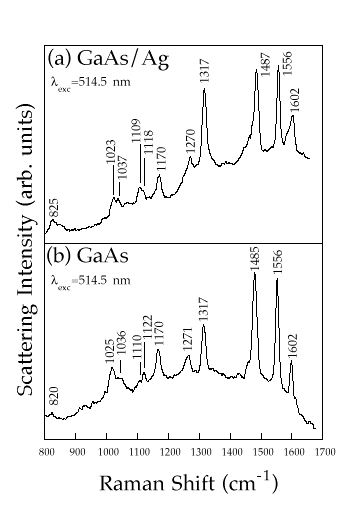
Figure 3. Raman spectra from [Ru(bpy)3]2+ adsorbed on: (a) GaAs/Ag system, (b) GaAs surface subjected to the etching treatment.
These results allow us to be confident that we have observed SERS spectra of [Ru(bpy)3]2+adsorbed on to GaAs surfaces coated with an Ag layer as well as subjected to a chemical etching treatment, in both cases the Raman enhancement has excellent time stability and reproducibility.
Conclusions
In conclusion, we show that SERS spectroscopy can detect and characterize ions and presumably molecules adsorbed onto semiconductor surfaces. We have observed the SERS spectra of molecules adsorbed on semiconductors subjected to a chemical etching process or coated with an island Ag film. Moreover, these results showed unequivocally that in the latter case the SERS signal arises from molecules adsorbed to GaAs surface and not to the Ag film itself .
This work is particularly significant from the SERS point of view because it shows that semiconductors are capable of supporting SERS.
Acknowledgments
We are grateful to B. Jusserand for many useful discussions and to F. R. Ladan for providing the silver depositions. We thank A. Ricci for technical assistance and P. Cafarelli for preparing the solution of [Ru(bpy)3]2+.
References
- R. K. Chang and T. E. Furtak, Surface Enhanced Raman Scattering Plenum: New York, 1982.
- A. Otto, Light Scattering in Solids IV, Topic in Applied Optics ed. by M. Cardona and G. Guentherodt, Springer, Heidelberg, 289 (1984).
- M. Moskovits, Rev. Mod. Phys. 57, 783 (1985).
- M. Fleischmannn, P. J. Hendra and A. J. Mac Quillan, Chem. Phys. Lett. 26, 163(1974).
- Q. J. Huang, X. Q. Li, J. L. Yao, B. Ren, W. B. Cai, J. S. Gao, B. W. Mao and Z. Q. Tian, Surface Science 427-428, 162 (1999).
- R. P. Van Duyne and J. P. Haushalter, J. Phys. Chem. 87, 2999 (1983).
- R. P. Van Duyne and J. P. Haushalter, J. Phys. Chem. 89, 405 (1985).
- V. L. Schlegel and T. M. Cotton, Anal. Chem. 63, 241 (1991)
- L. G. Quagliano, B. Jusserand and F. R. Ladan, physica status solidi (a) 152, 219 (1995)
- L. G. Quagliano and D. Orani, Surface Science 368, 108 (1996).
- H. Ishida, H. Fukuda, G. Katagiri and A. Ishitani, Applied Spectroscopy, 40, 322 (1986).
- G. Mattei, L. G. Quagliano and M. Pagannone Europhysics Letter 11, 373 (1990).
- N.L. Dmitruk, T.R. Barlas and E. V. Pidlisnyi, Surface Science 293, 107 (1993.
- P. G. Bradley, N. Kress, B. A. Hornberger, R. F. Dallinger and W.H; Woodruff, .J . Am. Chem. Soc. 103, 7441 (1981).
REF: L. G. Quagliano, Int. J. Vib. Spect., [www.irdg.org/ijvs] 4, 2, 3 (2000)