The application of Raman Spectroscopy to reacting systems
7. The application of Raman Spectroscopy to reacting systems
Andy Brookes and Derek Craston,
LGC, Queens Road,
Teddington,
Middlesex,
TW11 0LY
Abstract
The purpose of this paper is to outline applications for Raman spectroscopy in the field of reaction monitoring. The ability to monitor non-invasively, non-destructively and in real time makes Raman a particularly powerful technique in this area. Examples of real time monitoring are shown by the polymerisation of styrene and the curing of a commercial cyanoacrylate adhesive. Another case study shows the applicability of Raman to composite material degradation.
Introduction
Raman spectroscopy has been applied to an extremely wide variety of chemical systems. The ability to study solids, liquids, gases and solutions non-invasively and non-destructively combined with sampling through glass, optical fibres, microscopes and macro optical arrangements makes Raman an extremely versatile technique. Two excellent reviews cover many of the applications of Raman [1,2].Introduction
This paper is specifically concerned with reaction monitoring and aims to provide an indication of some of the uses of Raman in this field. The advantages gained from in situ process monitoring which have been facilitated by the increased use of fibre optic probes and chemometrics has made this an area of increasing importance. IR, NIR and to a lesser extent Raman and ultrasound are all beginning to make an impact as industry moves away from a reactive to a proactive attitude towards analysis.
To highlight its versatility Raman was applied to three different chemical systems.
Styrene Polymerisation
This liquid based (aqueous suspension of styrene) polymerisation reaction has been studied extensively using Raman [3]. Whilst both in situ[4] and flow cell [5] based systems have been used to study polymerisation reactions in the laboratory environment, the data presented here was obtained from a laboratory experimental set-up using a fibre optic probe. The work highlights the industrial applicability of information obtained from Raman experiments and also gives an indication of the large amount of data which can be easily collected when one avoids removing aliquots and working up the sample prior to analysis.
Why was Raman chosen to monitor this reaction? The desire for non-invasive, non-destructive real time monitoring limited the choice of analytical tool to spectroscopic techniques. The water present gives rise to problems in the NIR and IR due to its strong spectra but has a very weak Raman spectrum. The vinyl band, the functional group of interest, gives a very strong Raman signal making its disappearance easy to follow. This band is much weaker in the IR spectrum, making its analysis much more difficult (but not impossible). Also in the reaction mixture are additives e.g. initiators, although these are at low concentration there is a possibility that they could act as interference’s in some analytical methods. When using Raman their spectra are too weak to interfere with the styrene spectrum.
Adhesive Curing
Adhesive curing is an area where non-invasive sampling is highly desirable. Fluorescence has been a problem in the analysis of curing systems but the introduction of NIR sources has largely overcome this. Raman has been shown to be capable of monitoring the cure of several adhesive systems such as cyanoacrylate, methacrylate and epoxy [6]. This data has been used to evaluate the kinetics and mechanism of curing as well as evaluating individual components such as crosslinking agents [7].
Composite material degradation
As well as the curing of polymeric systems an important issue to manufacturers is degradation [8]. This is particularly important when considering composite materials. These are seen as the materials of the future, combining what are often very different materials to give desirable physical properties [9]. In recent years a wide array of composite materials has been developed. These present a new set of analytical challenges. The extent and cause of degradation of these materials and the effects the degradation has on the chemical and physical properties needs to be investigated thoroughly in order to ensure they are suitable for their proposed applications.
The main issue of interest in the analysis discussed here is the ingress of water into the material. Although the exact storage conditions and sample composition is not known it is believed that the samples were a polymer/glass composite and the storage conditions were the same for all of the samples with the only variable being time. Whilst it would seem sensible to study this problem with IR or NIR this was not feasible. As the material degrades it becomes darker in colour. The darkening of the material increases the IR and NIR absorbance of the material at all wavelengths preventing any meaningful IR and NIR analysis. Using visible excitation Raman spectra could not be collected due to fluorescence but FT Raman gave excellent spectra without problems from sample heating.
Instrumentation
Over recent years the number of instrument manufacturers has increased. Users are no longer restricted to a very limited number of specialist suppliers but can choose from a wide variety of vendors. Most of the new manufacturers have instruments built around diode lasers, CCD cameras and fibre probes. These range in price between $10K and $100K. It is obvious that compromises have to be made when designing an instrument at low cost. These become apparent as limitations in sensitivity, spectral resolution and range. It appears that instrument manufacturers who work in a particular area e.g. document examination or process monitoring, are beginning to recognise the potential of Raman and are designing instruments specific to their application. An extensive list of manufacturers can be found on the ‘Links’ page of this journal.
One point has to be made here: all of the instruments of the type described above are prone to fluorescence problems by their very nature. When these systems are applied to real industrial samples a clearer indication of their usefulness will develop. There are two practical alternatives available for overcoming fluorescence
- FT Raman, proven to be successful at avoiding fluorescence and easily coupled to fibre optics. These may be expensive in comparison to some of the lower cost instruments discussed above.
- UV laser excitation, this is expensive and is largely untried with industrial samples but has had a large amount of success with biological systems. Sample degradation problems may be encountered with UV lasers due to the large input of energy when using these wavelengths.
Experimental
Styrene polymerisation
The styrene data presented here was collected using a Perkin-Elmer 1760 FTIR modified for use as a Raman. A ~17m fibre optic probe was used to couple the spectrometer to the sample. The sample was contained within a glass bulb (approximate volume 1ml) which was pressurised to 10 bar. The polymerisation solution contained all of the additives which would be present in the industrial process, along with water which was used as a diluent. These conditions were chosen to mimic the industrial batch polymerisation process as closely as possible. The reaction was carried out at a variety of temperatures: 80 – 140 oC in 10 oC increments. A temperature ramped run was also carried out. The ‘ramp’ was a constant gradient temperature ramp between 80 and 140 oC, calculated using a model built from the isothermal runs, to be the optimum process condition.
Adhesive curing
The commercial cyanoacrylate based adhesive was cured between an aluminium coated glass slide and a glass cover slip. By focusing on the adhesive at the aluminium surface any interference from the spectrum of the glass slide was avoided. Spectra were obtained using a Renishaw Raman Imaging Microscope, at hourly intervals for 19 hr. The instrument used a CCD detector and laser excitation at 780 nm. The laser power was 5 mW, spectral resolution was 4cm-1 and spectra were acquired for 100 s between 3500 and 150 cm-1. A x50 objective was used. Data was obtained at room temperature only.
Composite material degradation
Composite material samples were obtained in various stages of degradation. Although it was attempted to obtain spectra using a CCD based system at 780 nm, fluorescence became a problem for the more degraded samples. Spectra were obtained successfully from a Perkin Elmer FT Raman System 2000 with laser excitation at 1064 nm and a quartz beamsplitter. Spectra were obtained from 16 scans between 200 and 3500 cm-1 with a laser power of 200 mW and a spectral resolution of 8 cm-1. Samples were labelled 1-4 in increasing state of degradation.
Results and Discussion
Styrene polymerisation
The spectra obtained from a styrene polymerisation reaction are shown in Figure 1. The spectra have been normalised to the intensity of the band at 1000 cm-1. It can be seen that whilst the intensity of this band, the breathing mode of the aromatic ring, is reasonably constant the vinyl band at ~1640 cm-1 decreases in intensity with time. The intensity of the vinyl band is directly proportional to the concentration of styrene monomer, and is plotted as % conversion (equation 1) against time for a variety of temperatures, Figure 2.
![]()
Equation 1
Where Mo is the monomer concentration at time 0 and Mt is the monomer concentration at time t. The area or intensity of the vinyl band can be substituted directly for Mo and Mt without having to calculate the monomer concentration.
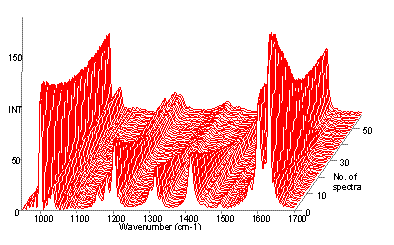
Figure 1. Raman spectra of the polymerisation of styrene, showing the disappearance of the vinyl band at ~1640 cm-1.
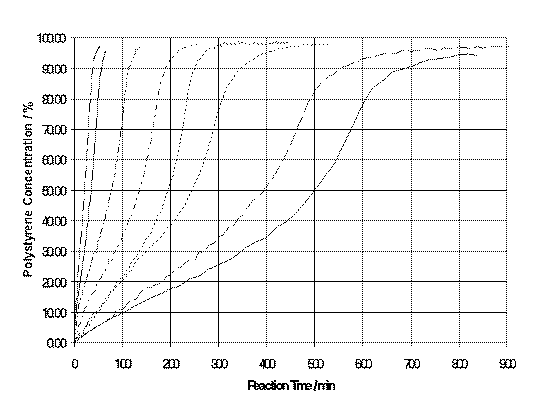
Figure 2. Plot of % conversion versus time for the polymerisation of styrene at(from right to left) 80, 90, 100, 110, calculated temperature ramp (80-140 oC in 250 min), 120, 130, 140 oC.
The data obtained from each isothermal polymerisation was placed into a kinetic model. The model was then used to optimise the process by calculating the best temperature ramp conditions. A temperature ramp is desirable as these conditions are known to result in the least residual monomer in the polymer. The calculated ramp conditions were also found to give the highest % conversion in the shortest time whilst yielding a polymer with appropriate physical properties e.g. molecular weight distribution.
Adhesive curing
On the basis of this laboratory work a Raman system was installed upon an industrial batch reactor to monitor the production of polystyrene. This is an excellent example of taking an analysis out of the laboratory and placing it into an industrial environment.
Figure 3 shows the spectra of a cyanoacrylate adhesive during curing. The spectra have been normalised to the CH aliphatic stretch region, 2960-2910 cm-1. It can be seen that many of the bands are decreasing in intensity with time. This seems to indicate that there may be several chemical changes occurring in the system. The intensity of several bands remains unchanged with time e.g. 1420, ~1000 and ~500 cm-1. The 1420 cm-1 band can be explained by the bending mode of CH2 or CH3. It appears likely that the bands at ~1000 and 500 cm-1 are due to C-C skeletal stretch and skeletal deformation respectively. The exact chemistry of this particular system is not known but it is perhaps unsurprising that these bands remain relatively constant when the spectra have been normalised to the aliphatic CH stretch region.
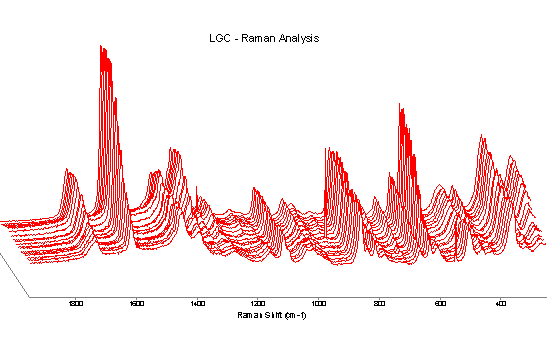
Figure 3. Curing of a cyanoacrylate adhesive. Spectra were obtained hourly for 19 hr (0hr at rear, 19hr at front).
Figure 4 shows the changes in the Cº N region over the course of the reaction. It can be clearly seen that the single Cº N stretch band present, 2235 cm-1, decreases in intensity as the reaction proceeds. Associated with this is the appearance and increase in intensity of the band at 2245 cm-1. It is likely that this is also due to CºN. This indicates either that the Cº N bond is being changed chemically, i.e. new substituents are being added to the carbon, or that identical groups are in two very different environments. The increase in intensity of the peak at 2245 cm-1 whilst the band at 2235 cm-1 is disappearing gives rise to an isosbestic point. This indicates that the two bands are very closely associated and dependent upon each other i.e. there is a one to one conversion from one form or environment to the other.
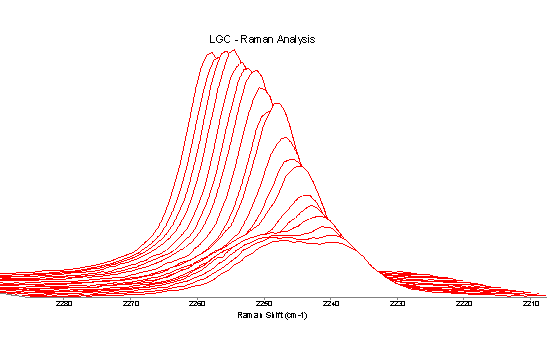
Figure 4. Raman spectra of the Cº N region showing the decrease in intensity of the band at 2235cm-1 and the appearance of the band at 2245cm-1 (0hr at rear and 19hr at front).
Figure 5 shows the changes in the region 900-700cm-1. There is a noticeable shift in position of the band at 860cm-1 whilst the band at 840cm-1 decreases in intensity greatly. Although there may be several assignments for the band at 840cm-1, it is possibly due to an oxirane ring (COC).
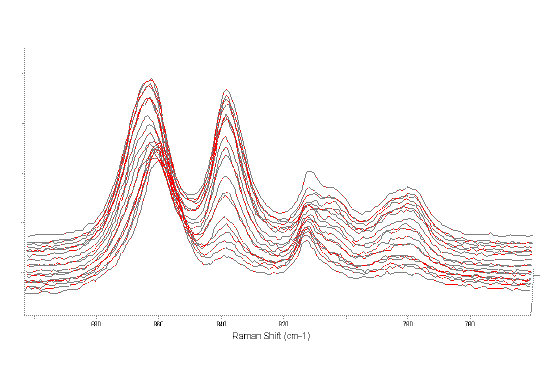
Figure 5. Showing the disappearance of the Raman band at 840cm-1 with time (0hr to rear, 19hr at front).
As well as obtaining information on the chemistry of the reaction it is also possible to obtain information on the extent of cure. Figure 6 shows the data obtained from the decrease in intensity of the 840 cm-1 band. The intensity was expressed as % conversion (equation 1) and plotted against time. Although data for this reaction was only obtained at RT it would have been possible to obtain data at a variety of temperatures, if desired, in order to elucidate kinetic information such as reaction rates and activation energies.
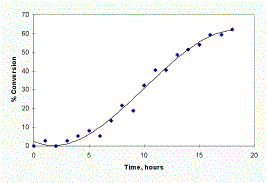
Figure 6. % conversion versus time for the band at 840 cm-1. The size of the symbols indicates the error in the Y axis. The increased % conversion at time 0-2hr is due to poor polynomial fit in this region and not to any experimental factor.
Composite material degradation
Figure 7 shows the spectra of composite material samples 1-4 (in increasing states of degradation). The increase in degradation was apparent from visual inspection. Although spectra 1 and 2 appear similar there is a clear difference between spectra 2 and 3. There is a more subtle difference between spectra 3 and 4.
It is apparent from the spectra that the material has some aromatic content. This is indicated (in the non-degraded spectra 1-2) by, for example, the ring breathing mode of the benzene ring at ~1000 cm-1 and the relatively intense aromatic CH stretch band, ~3050-3100 cm-1.
It is apparent that a large chemical change has occurred between samples 2 and 3. The ring breathing mode is no longer active and the relative intensity of the aromatic CH stretch has decreased. Also apparent is an increase in intensity of the OH stretch region (see expanded view, Figure 8) at ~3250-3500 cm-1.
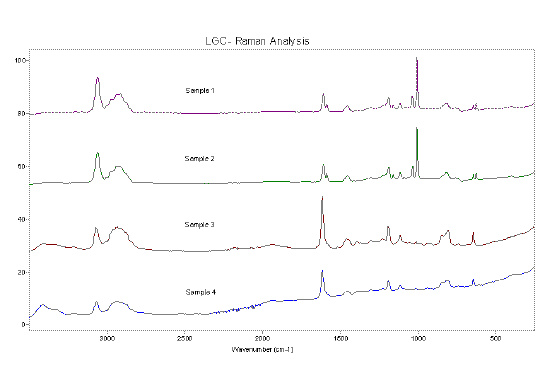
Figure 7. Spectra 1-4 showing a composite material in various states of degradation. A slight fluorescent background is visible in samples 3 and 4. Spectra are offset for clarity.
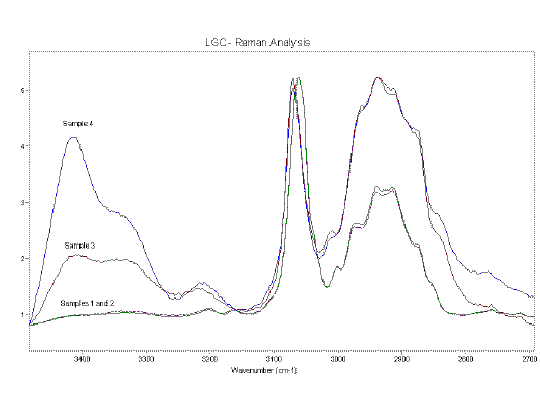
Figure 8. Raman spectra of the composite material samples showing the OH and CH stretch regions of the spectrum. All of the spectra are normalised to the aromatic CH stretch band, ~2950 cm-1.
Figure 8 shows that samples 1 and 2 have very similar spectra in the OH stretch (3250-3500 cm-1) and CH stretch region (2800-3100 cm-1), with very little contribution from OH. The difference between samples 2 and 3 are apparent. The aromatic CH stretch (3050 cm-1) to aliphatic CH stretch (2925 cm-1) ratio has dropped dramatically. There has also been an increase in the amount of OH (3250-3500 cm-1) present in the sample. Sample 4 has a similar aromatic to aliphatic CH stretch ratio but the amount of OH has increased above the level present in sample 3.
Associated with these changes are changes in the functional group and fingerprint regions of the spectra. Figure 9 shows these regions (1800-200 cm-1). The spectra show the change from the mono, meta or 1,3,5-substituted benzene ring pattern, spectra 1 and 2 (characterised by the intense band at ~1000 cm-1), to a pattern which is characteristic of other substitution patterns (characterised by the single intense band at ~1600 cm-1), spectra 3 and 4.
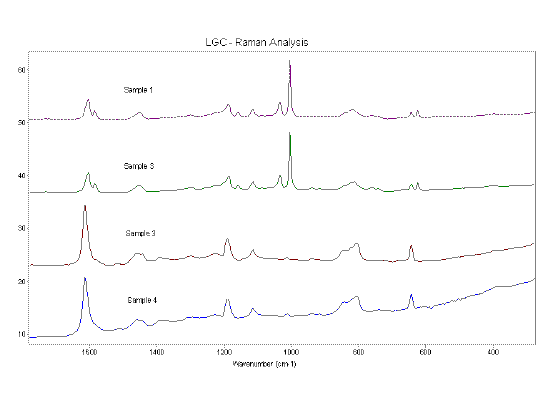 Figure 9. Composite material spectra between 1700 and 250 cm-1. An offset has been applied for clarity. A slight fluorescent background can be seen in samples 3 and 4.
Figure 9. Composite material spectra between 1700 and 250 cm-1. An offset has been applied for clarity. A slight fluorescent background can be seen in samples 3 and 4.
This evidence suggests that as the water content of the composite material increases the substitution on the aromatic ring also changes, associated with this is a decrease in the concentration of aromatic CH. This indicates an increase in substitution on the aromatic ring. The exact mechanism for this is unclear i.e. does the ingress of water into the material directly affect the substitution on the ring or does it facilitate another reaction in the material which gives rise to the change in substitution? It is also apparent from the differences between spectra 3 and 4 (Figure 8) that the OH concentration increases but the aromatic to aliphatic CH ratio stays the same, i.e. there is no further substitution on the aromatic ring. This may be due to a secondary reaction taking place which does not affect the aromatic to aliphatic ratio. There are no other changes in the spectra (samples 3 and 4) which are indicative of further chemical reactions. Another explanation is the retention of water by the material.
Although the mechanism for both the increased substitution on the aromatic ring and the further increase of the OH content of the material is unclear the effects upon the physical properties of the material are apparent e.g. severe discoloration of the material.
Conclusion
The styrene polymerisation data presented in this paper highlights the ability of Raman to obtain high quality chemical data from difficult sampling situations i.e. high temperature, high pressure, aqueous solutions. This work also illustrates the increased amount of data which can be obtained when aliquot collection is avoided. This means that continuous monitoring of processes or reactions can be achieved. Any errors associated with the work up procedure when using the aliquot method are also avoided. Also shown is the transferability of laboratory data to an industrial situation.
These issues also apply to the study of adhesive curing. It is possible to use Raman to study curing rates in order to establish curing kinetics and hence aid the elucidation of optimum product composition and ideal curing conditions.
The use of Raman to investigate degradation proved successful. Although full mechanisms could not be elucidated due to lack of information regarding the composition of the material, it appears that two mechanisms were taking place, one which changed the substitution on the aromatic rings and another which increased the OH content of the material without changing any of the chemical structure.
It is hoped that the elucidation of the degradation mechanisms of composite materials could aid in the discovery of alternative formulations which have greater resistance to degradation.
Acknowledgements
Elements of this paper were supported under contract with the Department of Trade and Industry as part of the Analytical Innovation Programme. Thanks also go to NPL for supplying the composite material samples and Actinic Technology for supplying the styrene polymerisation data.
References
- F.Adar, R.Gieger and J.Noonan, Appl. Spectrosc. Rev. 32, 45, 1997
- L.A.Lyon, C.D.Keating, A.P.Fox, B.E.Baker, L.He, S.R.Nicewarner, S.P.Mulvaney and M.J.Natan, Anal. Chem. 1998, 70, 341R-361R
- E.Gulari, K.McKiegue and K.Y.S.Ng, Macromolecules, 1984, 17, 1822
- J.Haigh, A.Brookes, P.J.Hendra, A.Strawn, C.Nicholas and M.Purbrick, Spectrochim. Acta Part A, 1997, 53, 9-19
- T.Ozpozan, B.Schrader and S.Keller, Spectrochim. Acta Part A, 1997, 53, 1-7
- M.Younes, S.Wartewig, D.Lellinger, B.Strehmel and V.Strehmel, Polymer, 1994, 35, 24, 5269
- J.K.F.Tait, H.G.M.Edwards, D.W.Farwell and J.Yarwood, Spectrochim. Acta Part A, 1995, 51, 2101-2106
- I.C.McNeill, Anal-Proc. 1983, 20, 11, 576-579
- C.J.DeBakker, G.A.George, N.A.StJohn and P.M.Fredericks, Spectrochim. Acta Part A, 1993, 49A, 5/6, 739-752
Received in original format 14th April 1999,
received in revised format and accepted 24th June 1999